IgG4 Class Switching, Immune Tolerance, and Adverse Event Risk from Repeated mRNA Booster Vaccination
Known Adverse Events, Mechanistic Risk Pathways, and Clinical Surveillance Priorities
IgG4 Class Switching, Immune Tolerance, and Adverse Event Risk from Repeated mRNA Booster Vaccination: Known Adverse Events, Mechanistic Risk Pathways, and Clinical Surveillance Priorities
This article was originally prepared in support of the ACIP Covid Working Group, and then formatted and submitted to the CDC journal MMWR (Morbidity and Mortality Weekly Report). With the recent court-ordered shutdown of the ACIP, I have pulled it and am now publishing it on Malone.News. The findings were summarized and scheduled for presentation at the ACIP public meeting, which was canceled on the order of the Boston district court and Judge Murphy. A simplified version of this information, with the slides developed for the ACIP presentation, was previously published on Malone.News
ABSTRACT
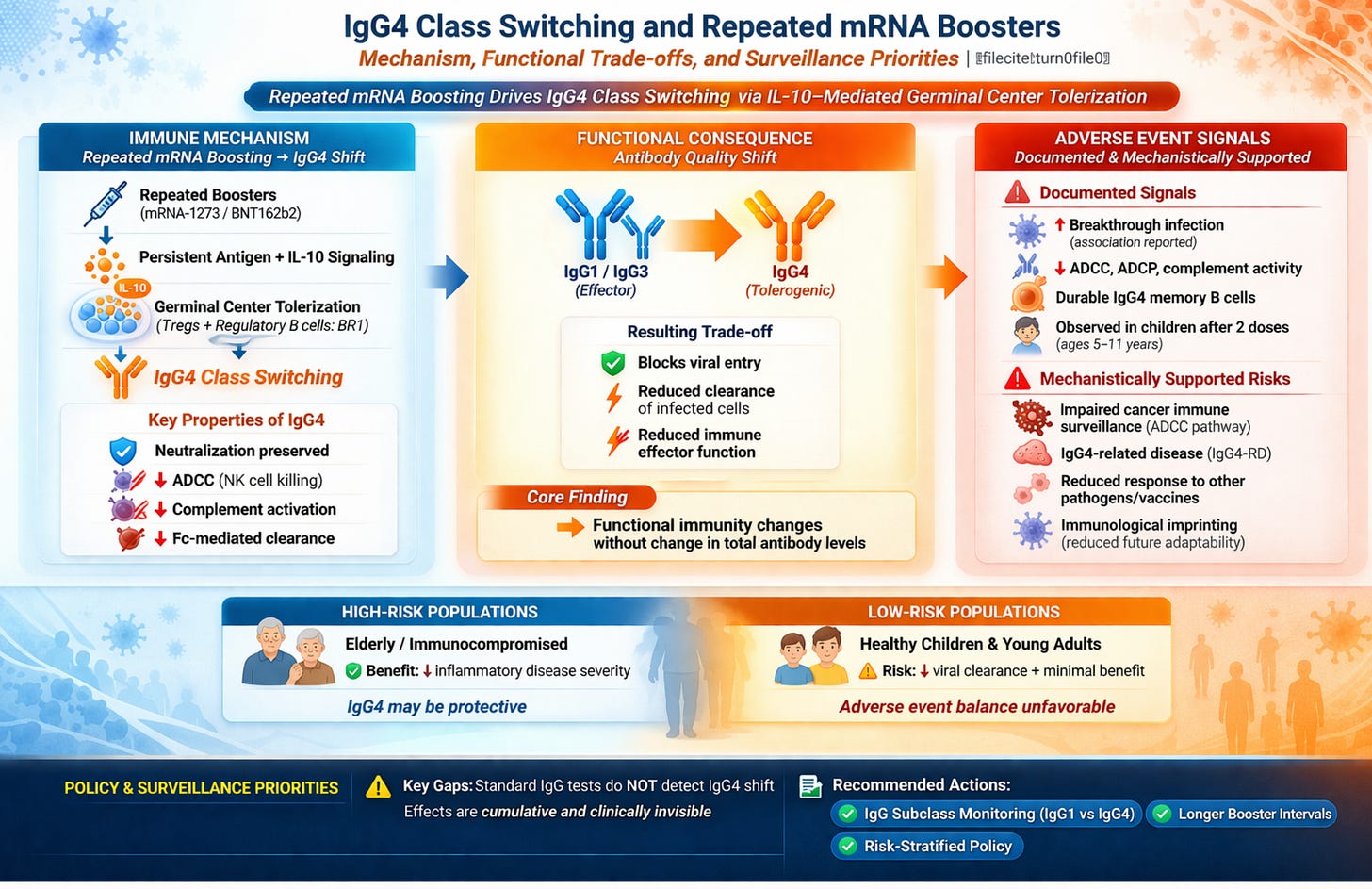
Background: Repeated COVID-19 mRNA booster vaccination drives a progressive IgG4 antibody class switch that carries documented and plausible adverse event risks warranting urgent clinical surveillance.
Methods: Systematic narrative review of peer-reviewed publications documenting IgG4 class switching following repeated mRNA vaccination, with analysis of immunological mechanism, concentration thresholds, functional consequences, and adverse event risk taxonomy.
Results: The class switch is mediated by IL-10 signaling within germinal centers, not systemic immunosuppression, but its consequences are durable: IgG4-expressing memory B cells and long-lived plasma cells encode a permanent change in the anti-spike antibody repertoire that manifests with every subsequent viral encounter. Documented adverse events include increased risk of SARS-CoV-2 breakthrough infection (Martín Pérez et al., J Infect 2025), impaired antibody-dependent cellular cytotoxicity (ADCC), complement activation failure, and loss of Fc-mediated viral clearance, confirmed by negative correlations between IgG4 levels and all three Fc effector functions simultaneously (r = −0.39 for neutralizing titers) (Kalkeri et al., J Infect 2025). Potential adverse event pathways with mechanistic support include: impaired immune surveillance of oncogenic pathogens and transformed cells secondary to Fc effector erosion; susceptibility to non-COVID pathogens dependent on IgG1/IgG3-mediated clearance; induction or exacerbation of IgG4-related disease (IgG4-RD) in predisposed individuals; generation of anti-idiotype and autoantibodies with pathological potential; and immunological imprinting that constrains protective breadth against future variant antigens. Pediatric-specific risk is documented: the class switch occurs in children aged 5–11 years after only two pediatric doses.
Conclusions: Standard anti-spike IgG serology cannot detect this shift, meaning adverse event risk is clinically invisible under current practice. Prospective adverse event surveillance incorporating IgG subclass assays, risk-stratified booster policy, and formal reassessment of pediatric booster indications are indicated.
Keywords: IgG4 class switching; COVID-19 mRNA vaccines; adverse events; immune tolerance; IL-10; IgG4-related disease; ADCC; Fc effector function; immunological imprinting; cancer immune surveillance; breakthrough infection; booster safety; pediatric vaccination; germinal center; regulatory B cells
1. IGG4 CLASS SWITCHING, IMMUNE TOLERANCE, AND THE BASIS FOR ADVERSE EVENT RISK
The unintended immunologic consequence of repeated COVID-19 mRNA boosting, now documented in multiple peer-reviewed publications, is progressive IgG4 class switching, occurring alongside sustained IL-10 signaling. This represents a qualitative shift in the humoral immune response from pro-inflammatory effector function toward immune tolerance. Understanding this shift, its mechanistic drivers, and its clinical significance is central to any rational assessment of universal booster policy.
1.1 Structural and Functional Properties of IgG4
IgG4 is considered tolerogenic because it preserves antigen recognition while actively limiting immune activation and inflammation. Among the four human IgG subclasses, IgG4 is the least abundant in healthy individuals and exhibits several structurally and functionally distinctive characteristics:
• Weak engagement of activating Fcγ receptors on immune cells, preventing amplification of inflammatory responses
• Inability to activate the classical complement pathway, blocking downstream inflammatory cascades
• Fab-arm exchange: IgG4 antibodies can swap one heavy-light chain pair with another IgG4 molecule, rendering many circulating IgG4 antibodies functionally monovalent, thereby preserving neutralizing activity while preventing effective cross-linking of the same antigen target
• Competition with IgG1 and IgE as a ‘blocking antibody,’ suppressing allergic and inflammatory reactions
• Active inhibition of antibody-dependent cellular cytotoxicity (ADCC), reducing elimination of SARS-CoV-2–infected cells
Together, these features allow IgG4 to recognize antigens (including the SARS-CoV-2 Spike protein) without provoking tissue-damaging inflammatory responses. IgG4 class switching is driven by regulatory immune environments rich in IL-10 and regulatory T cell (Treg) activity; it both reflects and reinforces anti-inflammatory immune regulation. Critically, IgG4 production is confined to human IL-10–producing regulatory B cells (BR1 cells) that potently suppress antigen-specific CD4+ T cell proliferation, meaning that the class switch itself is an active immunoregulatory event, not a passive consequence of antigen exposure alone.1
1.2 Peer-Reviewed Documentation of mRNA Vaccine-Induced IgG4 Switching
The following peer-reviewed publications document IgG4 class switching following repeated COVID-19 mRNA vaccination: post-vaccination IgG4 and IgG2 class switch associated with increased risk of SARS-CoV-2 infections2; class switch toward IgG2 and IgG4 more pronounced in BNT162b2 compared with mRNA-1273 vaccinees3; IgG4 class switching and decreased NK cell activation in older adults after repeated mRNA vaccination4; class switch toward IgG4 dependent on prior infection history5; IgG4 switching detected in children aged 5–11 years after two pediatric-dose vaccinations6; foundational longitudinal study documenting progressive IgG4 dominance after serial mRNA doses7; and a platform-comparison study confirming negative correlations between anti-spike IgG4 and neutralizing titers and all three Fc effector functions in the same cohort.8 [Note: reference 8 (Kalkeri et al.) was funded by Novavax; the majority of authors are Novavax employees. Findings should be interpreted with this conflict of interest in mind.]
1.3 Documented Adverse Consequences of IgG4 Dominance
The most immediate documented adverse consequence of IgG4 class switching is the functional erosion of the antiviral antibody response. IgG4 antibodies neutralize the virus: they block receptor binding and can prevent cell entry. What they cannot do is recruit immune effectors to eliminate already-infected cells. ADCC, the mechanism by which NK cells and macrophages recognize and destroy virus-infected cells coated with antibody, depends on IgG1 and IgG3 engaging activating Fcγ receptors, which IgG4 engages only weakly. Complement-mediated lysis of infected cells is similarly abolished. The result is an antibody that blocks viral entry at the cell surface but fails to clear the infection once established within tissues; therefore, cell-to-cell viral spread is not inhibited. This is not a theoretical trade-off: direct functional evidence confirms it. Anti-spike IgG4 levels are negatively correlated with neutralizing antibody titers (Spearman r = −0.39, p = 0.001) and with all three measured Fc effector functions—ADCP, ADCC, and ADCD—simultaneously, while IgG1 and IgG3 show positive correlations with the same endpoints.8 Critically, this negative correlation extends to neutralization itself, not just Fc effector functions: IgG4 accumulation does not add a tolerogenic layer on top of protective immunity but displaces the functional subclasses that mediate it. This displacement constitutes a clinically significant adverse event in patients who received repeated boosters under the assumption that rising anti-spike titer meant rising protection.
At the epidemiological level, one published study has directly associated the post-vaccination IgG4/IgG2 class switch with increased risk of subsequent SARS-CoV-2 breakthrough infection in a healthcare worker cohort.2 This constitutes the first formally reported adverse outcome associated with the IgG4 switch: increased susceptibility to the disease the vaccine was intended to prevent. This is an association requiring replication in larger prospective cohorts, and it does not establish that IgG4 switching causes the increased infection risk, as confounding remains possible. But the direction is mechanistically predicted, the signal is now in the published literature, and it should be treated as an adverse event signal warranting active pharmacovigilance. Immunologist Shiv Pillai provides an important calibrating perspective: IgG4 at current proportions is unlikely to abolish protection, given that IgG4 still neutralizes, crosses the placenta, and can form mixed complexes with IgG1. He explicitly acknowledges, however, that IgG4 dominance would meaningfully compromise Fc-mediated clearance, and his recommended interventions—annual minimum booster intervals, lower mRNA doses, heterologous protein subunit boosters—should be read as adverse event mitigation measures, not merely policy preferences.9
1.4 Comparison with Natural Infection
A critical distinction exists between the immune responses induced by natural SARS-CoV-2 infection and those produced by repeated mRNA vaccination. Natural infection predominantly induces IgG1 and IgG3 antibodies against the Spike protein, with little to no detectable IgG4 in convalescent individuals, a pattern consistent with classical antiviral inflammatory immunity. Primary mRNA vaccination similarly elicits strong IgG1 and IgG3 responses. However, with continued boosting, a pronounced class switch toward IgG4 becomes detectable months after the second dose and increases substantially after a third booster. This shift reflects prolonged antigen exposure and germinal center maturation following repeated vaccination, rather than an inherent feature of viral infection or initial immunization. The clinical implication is significant: individuals who have received multiple boosters may carry a qualitatively different antibody repertoire than those whose immunity derives primarily from infection or primary vaccination, one that is less well-equipped for Fc-mediated viral clearance despite comparable or higher total antibody titers. This divergence is not captured by standard serological assays and has not been accounted for in most comparative effectiveness analyses.
2. BOOSTER TIMING, SIGNAL STACKING, AND IGG4 INDUCTION
Booster timing determines whether immune signals resolve or accumulate (’stack’), and IgG4 class switching is most likely when boosting occurs before full inflammatory and germinal center resolution. This has critical mechanistic implications for booster scheduling policy.
2.1 Signal Stacking Mechanism
When boosters are closely spaced, residual antigen expression, ongoing germinal center activity, and unresolved innate signaling overlap with the next dose. This creates signal stacking: repeated exposure to the same antigen in a relatively low-inflammatory context. Under these conditions, regulatory feedback pathways—notably IL-10, regulatory dendritic cells, and Tregs—become progressively stronger, and B cells are increasingly likely to class-switch toward IgG4 rather than inflammatory subclasses such as IgG1 or IgG3. The immune system interprets the antigen as persistent but non-dangerous and adapts by dampening effector inflammation while preserving binding and neutralization.
2.2 Antigen Clearance and Interval Requirements
For pseudouridine-modified mRNA vaccines, antigen clearance may require many months due to enhanced mRNA stability conferred by nucleoside modification. Longer booster intervals allow antigen clearance, contraction of germinal centers, normalization of innate signaling, and resolution of regulatory cytokines before re-exposure. When a booster is administered after this reset, the immune system is more likely to re-engage inflammatory pathways and reinforce IgG1/IgG3 responses, reducing the probability of IgG4 dominance.
IgG4 class switching is generally not observed after primary vaccination or widely spaced boosts, but becomes more apparent after multiple, relatively frequent mRNA booster doses. This is why experimentally determined booster intervals, rather than reactive scheduling based on declining antibody titers, are essential for rational vaccine policy.
3. COVID-19 AS AN INFLAMMATORY DISEASE: IMPLICATIONS FOR VACCINE-INDUCED ADVERSE EVENTS
Coronavirus disease 2019 is an infectious disease in which host inflammatory responses are the primary determinant of severity and clinical outcome. Dysregulated or excessive inflammation, not direct viral cytopathic effects, drives the most serious complications: acute respiratory distress syndrome (ARDS), thromboinflammatory events, endothelial injury, and multisystem inflammatory syndromes. This inflammatory pathophysiology is directly relevant to understanding which vaccine-induced adverse events are most likely, in whom, and under what immunological conditions. A vaccine strategy that shifts antibody quality toward tolerogenic IgG4 will have different adverse event profiles in populations whose primary threat is inflammatory pathology versus those whose primary threat is viral replication. These profiles have not been prospectively characterized, and the absence of prospective data does not mean the absence of adverse events; it means they have not been systematically looked for.
Clinical severity correlates more strongly with markers of systemic inflammation than with viral burden beyond the early phase of infection. Complications including ARDS, thromboinflammatory events, endothelial injury, and multisystem inflammatory syndromes are predominantly mediated by immune and inflammatory pathways rather than direct viral cytopathic effects. This understanding has important implications for interpreting the immunologic effects of mRNA vaccines and for designing population-specific vaccination strategies.
3.1 Population-Specific Adverse Event Risk Profiles
The bimodal nature of COVID-19 pathophysiology produces two distinct adverse event risk profiles when IgG4 class switching is superimposed on population-specific disease patterns:
• High-risk populations (elderly, immunocompromised, metabolically unhealthy): Characterized by elevated inflammatory set points predisposing to pronounced secondary inflammatory responses. For these groups, reducing morbidity and mortality from inflammatory disease sequelae is the primary endpoint. The tolerogenic properties of IgG4 may be adaptive and protective.
• Low-risk populations (healthy children and young adults): At minimal risk for severe disease or death but central to viral spread and community transmission. For these groups, reduction in viral replication and spread should be the primary objective, a goal that IgG4-dominant tolerogenic immunity is ill-suited to fulfill.
A universal booster policy does not acknowledge these divergent adverse event profiles and cannot address them. For elderly and immunocompromised patients, the tolerogenic shift may reduce the inflammatory adverse events most dangerous to them (cytokine storm, ARDS, thromboinflammatory sequelae) while preserving neutralization. For this group the adverse event calculus may favor continued boosting with interval discipline.
For healthy children and young adults who face negligible individual risk of severe disease, the adverse events associated with repeated boosting are different in character: progressive erosion of Fc effector function with no offsetting reduction in severe disease risk; immunological imprinting that constrains future vaccine and infection responses; and documented IgG4 switching after only two pediatric doses. In this group the adverse event calculus is distinctly unfavorable, and the claim that repeated boosting is straightforwardly beneficial is not supported by the available evidence.
4. ADVERSE EVENT: ALTERED IMMUNE SELECTION PRESSURE AND SARS-COV-2 EVOLUTION
A consequence of widespread IgG4 class switching that has received insufficient attention as an adverse event is its potential contribution to SARS-CoV-2 immune escape evolution. Viral evolution is overwhelmingly driven by replication frequency and selective immune pressures operating at the population level. A vaccination strategy that induces IgG4-dominant antibody responses, maintaining neutralization pressure while eliminating Fc-mediated clearance, creates a specific and potentially novel immune selection environment. Viruses that escape neutralization are eliminated; viruses that are neutralized but persist in the presence of deficient Fc effector activity can replicate, spread, and evolve. The long-term adverse consequence is a viral fitness landscape increasingly shaped by pressure from a single, Fc-depleted immune mechanism, potentially accelerating the emergence of neutralization-escaping variants while leaving T cell and inflammatory immune targets under less selection pressure than a balanced IgG1/IgG3/IgG4 response would produce.
Evidence strongly supports the conclusion that SARS-CoV-2 evolution has been driven primarily by selection for neutralization escape. One study integrating deep mutational scanning, antibody neutralization data, and genomic surveillance demonstrated that SARS-CoV-2 variants continually acquire spike mutations reducing susceptibility to neutralizing antibodies, with immune escape dynamics matching historical variant prevalence and fitness trends.10 A second study demonstrated that spike mutations conferring resistance to commonly elicited neutralizing antibodies can be readily selected in vitro, supporting neutralization escape as a key evolutionary driver.11
SARS-CoV-2 evolution shows strong evidence of selection for escape from neutralizing antibodies, with mutations concentrated in spike protein epitopes, while targets of inflammatory and T-cell–mediated immunity remain largely conserved. This pattern indicates viral fitness is driven by evasion of neutralization to sustain transmission in partially immune populations, a dynamic that tolerance-oriented immune profiles may inadvertently facilitate.
5. IL-10: MECHANISM, CONCENTRATION THRESHOLDS, AND CLINICAL SIGNIFICANCE
IL-10 is a pleiotropic cytokine with complex, context-dependent, and concentration-dependent effects on the immune system. A rigorous assessment of its role in repeated mRNA vaccination requires careful separation of three distinct questions: (1) what IL-10 concentrations are produced by repeated boosting; (2) at what concentrations IL-10 produces clinically meaningful immune effects; and (3) whether the mechanism driving IgG4 class switching requires systemic IL-10 elevation at all. The answers to these questions substantially reshape the clinical significance argument.
5.1 IL-10 Reference Ranges and Booster-Associated Levels
Multiple clinical reference ranges establish healthy adult serum IL-10 at less than 3–10 pg/mL depending on the assay platform. One study of 50 normal volunteers found a median IL-10 level below 3.0 pg/mL.12 Another found healthy adult serum IL-10 ranging from 4.8–9.8 pg/mL (mean 7.1 pg/mL), using ≥10 pg/mL as the threshold for clinical elevation.13 Clinical laboratory reference ranges treat values in this range as normal, with meaningful elevation defined operationally as a two- to several-fold increase above baseline.
Published cytokine data following COVID-19 mRNA booster doses document modest, transient elevations in this range. Rosati et al. measured serum cytokines after the third BNT162b2 dose and found IL-10 induced transiently, clustering with IL-27 in a regulatory cytokine module alongside a broader inflammatory signature that persisted up to one month post-dose.14 A Japanese longitudinal study of frail elderly individuals found that IL-10 levels three months post-booster were negatively associated with indicators of frailty including Eastern Cooperative Oncology Group (ECOG) performance status and serum albumin.15 Importantly, no large longitudinal human study has directly quantified absolute serum IL-10 across multiple booster doses using a standardized platform. Most published data report fold-changes from baseline rather than absolute pg/mL values, and inter-assay comparability is poor.
The critical implication: transient booster-induced IL-10 elevations, as currently documented, remain in the low pg/mL range. This is insufficient to produce broad systemic immunosuppression. The hypothesis that repeated boosting causes meaningful systemic immune compromise via IL-10 elevation alone is not yet supported by direct human data and should be clearly labeled as mechanistic inference. The clinical significance of booster-associated IL-10 lies elsewhere: in the local tissue microenvironment, not in systemic cytokine concentrations.
5.2 Concentration Thresholds for Clinically Significant Immune Effects
A substantial body of disease-state literature defines what constitutes clinically meaningful IL-10 elevation. These data provide essential context for calibrating the significance of vaccine-induced changes.
In oncology, IL-10 levels above 10 pg/mL are associated with inferior failure-free survival in Hodgkin’s disease13; levels above 169 pg/mL predict poor prognosis in multiple myeloma16; and levels above 4,300 pg/mL (4.3 ng/mL) are associated with significantly shorter progression-free and overall survival in renal cell carcinoma treated with immune checkpoint inhibitors.17 Melanoma patients exhibit median serum IL-10 of 8.75 pg/mL versus less than 3.0 pg/mL in healthy controls, with levels above 10 pg/mL associated with worse survival.12 In acute coronary syndromes, IL-10 levels above 3.5 pg/mL were associated with reduced cardiac risk, reflecting IL-10’s anti-inflammatory cardioprotective role in acute inflammation.18
In vitro studies of NK cell function provide perhaps the most informative pharmacodynamic data. NK cell cytotoxicity and IFN-γ secretion are significantly enhanced as IL-10 concentrations increase from 5 to 50 ng/mL—that is, pharmacological concentrations three to four orders of magnitude above normal serum levels.19,20 The half-maximal effective concentration of pegylated IL-10 in cell-based assays is approximately 8 ng/mL, while baseline serum IL-10 in cancer patients (who have elevated levels) averages around 3 pg/mL.21 At concentrations in the high-ng/mL range used therapeutically, IL-10 paradoxically enhances CD8+ T cell and NK cell cytotoxicity, an effect that is entirely irrelevant to the pg/mL range produced by vaccination. At lower concentrations, IL-10’s suppressive effects on dendritic cells and antigen presentation are documented and mechanistically important, but the precise thresholds in human tissue remain poorly defined.
The conclusion is that the serum IL-10 elevations attributable to repeated COVID-19 mRNA booster vaccination are far below the concentrations documented to produce systemic immunosuppression, impair NK cell cytotoxicity, or reduce CD8+ T cell responses in clinical disease states. Any claim that booster vaccination produces broad systemic immunosuppression via elevated serum IL-10 is not supported by current quantitative evidence and represents an overreach that should be explicitly disavowed.
5.3 The Germinal Center Mechanism: Where IL-10 Clinical Significance Is Located
The foregoing concentration analysis does not diminish the clinical significance of booster-associated IL-10; it precisely locates it. Booster-associated IL-10 is not producing systemic immunosuppression detectable in the bloodstream. It is producing a targeted, local immunoregulatory event within germinal centers that directly and permanently reshapes the antibody repertoire. This distinction matters clinically because it means the harm is not diffuse and recoverable (as elevated systemic IL-10 would be when it clears) but encoded into the B cell memory pool: once IgG4-expressing memory B cells and long-lived plasma cells are generated, they persist in bone marrow for years and continue to produce IgG4 upon antigen re-encounter. Each additional booster dose reinforces and expands this pool. The immunologically meaningful effects of IL-10 on IgG4 class switching operate at the level of local tissue signaling within germinal centers and antigen-experienced B cell microenvironments, but their clinical consequences manifest every time the patient is re-exposed to SARS-CoV-2.
IgG4 production is selectively confined to human IL-10–producing regulatory B cells (BR1 cells).1 In B cell cultures, IL-10 increases IL-4–induced IgG4 production more than 20-fold, acting directly on antigen-experienced, switched B cells in tissues where IL-10 receptor expression is higher.22 This direct action does not require systemic IL-10 elevation; it requires only that an IL-10–rich microenvironment exists within germinal center or memory B cell niches at the time of antigen re-encounter.
This mechanism is well-established in the allergen immunotherapy literature, where repeated high-dose antigen exposure drives IgG4 switching via IL-10–producing Tregs and regulatory B cells. IgG4 production is confined to IL-10+ BR1 cells in vivo, and beekeepers tolerant to bee venom exhibit high IgG4/IgE ratios corresponding to increased frequencies of allergen-specific IL-10–producing B cells.1,23 IL-10 inhibits IgE production and enhances IgG4 production, changes that coincide with clinical tolerance.24 The COVID-19 mRNA vaccine context reproduces the key immunologic conditions for this switch: repeated high-dose antigen (Spike protein) exposure in an environment where each booster further reinforces IL-10–driven regulatory signaling before the preceding response has fully resolved.
The clinical significance of booster-associated IL-10 is therefore not systemic immunosuppression but rather germinal center tolerization: a progressive shift in the quality of the antiviral humoral immune response toward blocking antibodies with reduced Fc effector function. This is a clinically meaningful distinction: IgG4 preserves neutralization but eliminates ADCC, complement activation, and inflammatory viral clearance. At current IgG4 proportions, this likely reduces the breadth and quality of antiviral protection without causing generalized immune dysfunction. Direct functional evidence for this trade-off has now been reported: in a platform-comparison study, anti-spike IgG4 levels were negatively correlated with neutralizing antibody titers (Spearman r = −0.39, p = 0.001) and with all three measured Fc effector functions (ADCP, ADCC, and ADCD) while IgG1 and IgG3 showed positive correlations with the same endpoints.8
5.4 Potential Adverse Event: Impaired Immunity to Non-COVID-19 Pathogens
A potential adverse event pathway that has not been prospectively studied is impaired immune responses to unrelated pathogens and vaccines following repeated mRNA booster vaccination. The IgG4 class switch is spike-specific in its immunological induction, but its consequences may extend beyond anti-spike responses. IgG4-expressing regulatory B cells (BR1 cells), which produce IL-10 and suppress antigen-specific T cell proliferation, are expanded by repeated booster vaccination. Whether this expansion alters the regulatory tone of the immune system in ways that affect responses to co-presented or subsequently encountered antigens is an open question with genuine clinical stakes.
Analogous to findings in helminth infections, where chronic IL-10 elevation shifts both vaccine and infection responses toward anti-inflammatory, tolerance-promoting profiles, repeated mRNA vaccination might reduce immunogenicity for vaccines relying on strong Th1 signaling, or impair clearance of pathogens that normally depend on IgG1/IgG3-mediated effector functions. In aged mice, IL-10–secreting Tfh10 cells suppress immune responses and contribute to vaccine non-responsiveness reversible by IL-10 neutralization.25 Whether repeatedly boosted humans exhibit similar non-specific immune modulation is unstudied in prospective cohorts. This is not a demonstrated adverse event. It is a plausible adverse event pathway that has not been ruled out and that demands prospective investigation, particularly given that the populations most heavily boosted are also the populations most likely to be concurrently receiving other vaccines.
5.5 Potential Adverse Event: Impaired Cancer Immune Surveillance
Cancer immune surveillance is a legitimate adverse event concern in the context of repeated mRNA booster vaccination, but the mechanistic pathway that matters is not systemic IL-10 elevation but rather ADCC impairment and NK cell functional erosion. These are distinct claims that require separate evaluation. The IL-10 pathway: elevated serum IL-10 exerts tumor-promoting effects through STAT3 activation, Treg recruitment, HLA class I downregulation, and NK cell suppression.26 However, these effects are documented in disease states characterized by sustained substantially elevated IL-10, not the transient low-pg/mL elevations produced by vaccination. At pharmacological concentrations used therapeutically, IL-10 paradoxically enhances NK cell and CD8+ T cell cytotoxicity.19,21 The cancer surveillance concern from vaccine-induced systemic IL-10 cannot currently be substantiated by quantitative data and should not be overstated.
The ADCC pathway is a different and more credible adverse event concern. ADCC is a primary mechanism of cancer immune surveillance: NK cells bearing FcγRIII (CD16) recognize tumor cells coated with IgG1 or IgG3 antibodies directed against tumor-associated antigens and eliminate them. IgG4 does not support this mechanism. Multiply-boosted individuals in whom the balance of their total IgG response has shifted toward IgG4 may have reduced ADCC capacity not only against SARS-CoV-2–infected cells but potentially against any target cell recognized by antibodies whose Fc region is competed by high circulating IgG4. The degree to which spike-specific IgG4 expansion alters the functional Fc receptor engagement capacity of the broader antibody repertoire is not directly characterized in the available literature. The plausibility of this mechanism is supported by the well-documented IgG4-mediated reduction in ADCC against spike-positive cells,8 by NK cell functional impairment documented in repeat-boosted older adults,4 and by the known biology of IgG4 as a competitive inhibitor of activating Fc receptor engagement. This adverse event pathway is mechanistically supported but not yet quantified in terms of clinical outcome risk. It warrants prospective study, particularly in patients with active or recent malignancy who depend on intact NK cell-mediated surveillance.
A further adverse event pathway requiring explicit discussion is IgG4-related disease (IgG4-RD). IgG4-RD is a systemic fibro-inflammatory condition characterized by tissue infiltration of IgG4-positive plasma cells, storiform fibrosis, and elevated serum IgG4, affecting the pancreas, bile ducts, salivary glands, orbits, kidneys, and other organs. Its pathogenesis involves dysregulated IgG4 production in a context of chronic antigen stimulation and IL-10-driven regulatory signaling, the same immunological substrate that repeated mRNA booster vaccination systematically reinforces. The question of whether vaccination-driven IgG4 expansion in predisposed individuals can precipitate or exacerbate IgG4-RD has not been prospectively studied. Individual case reports of IgG4-RD onset or flare following COVID-19 vaccination have appeared in the literature, though no systematic pharmacovigilance data are available. The biological plausibility of this adverse event pathway is high: the induction conditions overlap substantially, and individuals with subclinical IgG4-RD or genetic predisposition to regulatory B cell expansion represent a population who may be at disproportionate risk. This should be an explicit surveillance priority in post-marketing adverse event reporting frameworks.
6. KNOWN AND POTENTIAL ADVERSE EVENTS: CONSOLIDATED SUMMARY AND SURVEILLANCE PRIORITIES
This section consolidates the adverse event evidence reviewed across Sections 1–5 into a structured taxonomy distinguishing documented adverse events (with published epidemiological or functional evidence), mechanistically supported adverse event pathways (biologically plausible, not yet epidemiologically confirmed), and areas of genuine scientific uncertainty. This taxonomy is intended to support adverse event surveillance design and to resist two common epistemic failures in this literature: overclaiming harm without quantitative support, and dismissing plausible risk pathways because prospective confirmation is absent.
6.1 Documented Adverse Events
The following adverse events have published functional or epidemiological evidence directly linking repeated mRNA booster vaccination to a clinically adverse outcome. They do not yet meet the evidentiary standard of replicated randomized trial data, but they meet the standard for pharmacovigilance action: mechanistic plausibility combined with at least one published signal in peer-reviewed literature.
Increased susceptibility to SARS-CoV-2 breakthrough infection. Martín Pérez et al.2 documented a statistically significant association between the post-vaccination IgG4/IgG2 class switch and increased risk of subsequent SARS-CoV-2 infection in a healthcare worker cohort. This is the most direct adverse event signal currently in the literature and is mechanistically consistent with the Fc effector function impairments confirmed in the same period by Kalkeri et al.8 Causation is not established; the association requires replication. It should be entered into pharmacovigilance databases and used to trigger prospective cohort studies with IgG subclass stratification.
Loss of ADCC, complement activation, and Fc-mediated viral clearance. Documented directly in surrogate effector function assays by Kalkeri et al.8 and Irrgang et al.,7 and in NK cell activation assays in older adults by Ravichandran et al.4 Anti-spike IgG4 levels are negatively correlated with ADCP (r = −0.40), ADCC (r = −0.53), and ADCD (r = −0.53) simultaneously in the same cohort. These are not theoretical predictions but published measurements. The clinical consequence is reduced capacity to clear SARS-CoV-2–infected cells by immune effector mechanisms that operate independently of viral entry blocking, and this constitutes the mechanistic basis for the breakthrough infection adverse event above.
Durable B cell memory encoding functional impairment. Irrgang et al.7 demonstrated that IgG4-class-switched memory B cells constitute a median of 14.4% of all spike-binding memory B cells in the blood of triply-boosted healthcare workers. Long-lived plasma cells producing IgG4 are generated in germinal centers and persist in bone marrow for years, continuing to secrete IgG4 upon every antigen re-encounter. This is not a transient adverse event: the immunological impairment is written into the B cell memory pool in a manner that standard serology cannot detect and that persists independently of further vaccination. This constitutes a durable adverse change in the humoral immune repertoire.
IgG4 class switching in children after two pediatric doses. Kobbe et al.6 confirmed IgG4 class switching in children aged 5–11 years at one year after only two 10-μg BNT162b2 doses. Confounded by intervening Omicron breakthrough infection in all subjects, this study nevertheless establishes that the switch is not age-restricted and does not require three or more doses. The adverse event implications for children are particularly significant because the individual risk-benefit ratio for pediatric boosting against severe COVID-19 is negligible, meaning any adverse event, however small in absolute magnitude, carries disproportionate weight against the claimed benefit.
6.2 Mechanistically Supported Adverse Event Pathways
The following adverse event pathways are biologically plausible on the basis of established immunological mechanisms and the documented IgG4 switching data, but lack prospective epidemiological confirmation in repeatedly-boosted human cohorts. They are not demonstrated harms; they are risk pathways that pharmacovigilance and prospective research should actively investigate.
IgG4-related disease (IgG4-RD) precipitation or exacerbation. IgG4-RD is a fibro-inflammatory syndrome driven by the same IL-10–dominated regulatory B cell environment that mRNA boosters systematically reinforce. The induction conditions overlap substantially. Individual case reports of post-vaccination IgG4-RD flare exist in the literature; no systematic pharmacovigilance analysis has been conducted. Individuals with subclinical IgG4-RD, a history of the condition, or genetic predisposition to regulatory B cell hyperactivation may be at materially elevated risk from repeated boosting.
Anti-idiotype and autoantibody generation. The Spike protein shares structural and sequence homology with several human proteins. Repeated high-dose Spike antigen exposure in the context of prolonged germinal center activity and high somatic hypermutation rates (documented to be 3.5-fold elevated over six months in mRNA-LNP–induced GC reactions27) creates conditions favorable to the generation of cross-reactive antibodies with autoreactive potential. Anti-idiotype antibodies generated against anti-spike antibodies may additionally mirror the ACE2 receptor, with potential pathological consequences. The clinical relevance of this pathway in repeatedly-boosted individuals has not been systematically characterized, but the mechanistic conditions for it are well-established.
Impaired cancer immune surveillance via ADCC erosion. NK cells eliminate tumor cells through ADCC, recognizing IgG1/IgG3-coated transformed cells via FcγRIII. Multiply-boosted individuals in whom total anti-spike IgG includes a substantial IgG4 fraction show reduced ADCC capacity against spike-positive cells8 and reduced NK cell activation by spike-specific antibodies.4 Whether this Fc effector erosion extends to non-spike cancer surveillance targets is unknown; IgG4 circulates as a competitor for Fc receptor engagement and may reduce the effective ADCC capacity of IgG1/IgG3 antibodies against tumor-associated antigens in a bystander fashion. This adverse event pathway is supported by known IgG4 biology but not quantified in clinical oncology data from boosted populations. Patients with active or recent malignancy receiving checkpoint inhibitors or ADCC-dependent antibody therapies represent a high-priority monitoring population.
Interference with therapeutic monoclonal antibody efficacy. Many cancer immunotherapies (including rituximab, trastuzumab, cetuximab, and others) rely on IgG1-mediated ADCC and ADCP as primary or co-primary mechanisms of action. High circulating IgG4 can competitively occupy FcγRIII on NK cells and macrophages, reducing the availability of activating Fc receptors for therapeutic IgG1 antibodies. Whether booster-associated IgG4 expansion reaches concentrations sufficient to meaningfully blunt therapeutic antibody efficacy is not studied. This is an adverse event pathway with direct clinical drug interaction implications that should be examined in oncology patients receiving booster doses concurrent with ADCC-dependent therapies.
Impaired immunity to unrelated pathogens. Expansion of IL-10–producing regulatory B cells (BR1 cells) by repeated mRNA boosting may alter the regulatory tone of immune responses to non-spike antigens. Chronic helminth infections produce a comparable regulatory B cell expansion, with documented downstream effects on vaccine immunogenicity and pathogen clearance. Whether repeated COVID-19 boosters similarly reduce immunogenicity of co-administered influenza, pneumococcal, shingles, or other vaccines in the same individuals has not been studied in prospective controlled cohorts. This is an adverse event pathway of particular concern in elderly and immunocompromised patients who receive multiple vaccines simultaneously.
Immunological imprinting as a durable adverse event. Kim27 synthesized evidence that ancestral-spike memory B cells primed by primary mRNA vaccination outcompete naïve B cells upon re-exposure to variant antigens, constraining the breadth of responses to updated boosters and limiting variant-specific de novo antibody generation. This immunological imprinting is a durable adverse consequence of the prolonged germinal center activity induced by mRNA-LNP platforms: the same GC persistence that enables superior affinity maturation simultaneously encodes a structural constraint on future humoral adaptability. In practical terms, repeatedly-boosted individuals may respond less robustly to novel variant antigens and potentially to future pandemic pathogens than individuals whose humoral immunity derives from natural infection or primary vaccination. This is an adverse event whose full clinical magnitude cannot be known until a sufficiently divergent future pathogen or variant is encountered.
6.3 Adverse Event Surveillance Priorities
None of the documented or mechanistically supported adverse events reviewed in this section are captured by current post-marketing surveillance systems, because those systems are designed to detect symptomatic adverse events in the short-term following vaccination. The IgG4 class switch and its consequences are immunological, progressive, cumulative, and detectable only by assays not used in routine clinical practice or pharmacovigilance.
The following constitute minimum surveillance priorities: (1) Prospective cohort studies stratifying COVID-19 outcomes by IgG subclass composition in multiply-boosted individuals, with IgG4/IgG1 ratio as a primary analytical variable. (2) Systematic pharmacovigilance review of IgG4-RD case reports and case series with temporal association to COVID-19 vaccination. (3) Oncology registry linkage studies examining outcomes in cancer patients receiving ADCC-dependent therapies who also received multiple COVID-19 boosters. (4) Immunogenicity studies of co-administered vaccines in multiply-boosted versus primary-vaccinated individuals, with IgG subclass and NK cell functional assays as secondary endpoints. (5) Pediatric-specific studies with IgG subclass outcomes, examining children who received primary series vaccination versus those who received additional boosters, with long-term follow-up for immune competence markers. The absence of this data is not evidence of safety; it is evidence of an underpowered surveillance system that was not designed to detect the category of adverse event most likely to emerge from the IgG4 switching mechanism.
7. PLATFORM-SPECIFIC IMMUNOLOGY: MRNA VERSUS DNA VACCINES
7.1 Role of Pseudouridine Modification
Pseudouridine (Ψ) or N¹-methyl-pseudouridine (m¹Ψ) modification of synthetic mRNA attenuates acute innate inflammatory signaling by reducing activation of RNA sensors including TLR7/8 and RIG-I, thereby decreasing acute type I interferon responses while enhancing mRNA stability and antigen expression. This modification does not directly induce IL-10 production or IgG4 switching; rather, it creates an immune environment in which regulatory responses can emerge over time with repeated dosing. Prolonged antigen presentation and reduced acute inflammatory ‘danger’ signals support sustained germinal center activity, during which IL-10 may be upregulated as part of normal feedback control. Critically, the reduced innate inflammatory signal shifts the probability of IgG4 class switching upward by reducing the competing IgG1/IgG3-promoting inflammatory milieu.
7.2 DNA Contamination and cGAS-STING Signaling
Residual DNA fragments, produced during the mRNA manufacturing process, have been found in final drug products. These DNA fragments are DAM and DCM-methylated and may combine with hypermethylated (m¹Ψ) synthetic RNA to form dual-methylated RNA:DNA hybrids with no natural analog. Residual DNA can activate the cGAS-STING pathway. While acute STING signaling promotes antiviral and inflammatory immunity, prolonged or repeated activation induces counter-regulatory mechanisms including upregulation of IL-10, induction of regulatory dendritic cells and macrophages, expansion of Tregs, and functional exhaustion of effector T cells. This sustained cGAS-STING activation may represent an additional, compounding driver of the IL-10–rich, tolerance-promoting immune environment observed with repeated mRNA boosting.
7.3 Comparison with DNA Vaccine Platforms
DNA vaccines are generally associated with more strongly Th1-biased immune responses, characterized by higher relative induction of IgG1 and IgG3 antibodies and greater emphasis on cellular immunity. Innate sensing of DNA vaccines is driven primarily through the cGAS-STING axis in an acute, interferon-dominant context. Compared with mRNA platforms, DNA vaccines typically result in lower, shorter-term antigen expression and have been administered less frequently. There is limited evidence for IgG4 dominance following DNA vaccination, and IgG4 class switching has not been a prominent or reproducible feature of DNA vaccine platforms.
8. DISCUSSION
The adverse event taxonomy developed in Section 6 has a common structural feature that makes it acutely difficult to manage within existing pharmacovigilance frameworks: every adverse event pathway reviewed is immunological, progressive, cumulative, and detectable only by assays that are not used in routine clinical practice. Millions of people who received multiple COVID-19 mRNA boosters now carry an antibody repertoire whose functional character is meaningfully different from what standard serology implies. Total anti-spike IgG titer, the measure used to authorize boosting and to assess protection in clinical trials and real-world effectiveness studies, does not distinguish between subclasses.
A patient with high anti-spike IgG after a fourth or fifth booster may carry a response in which a substantial and growing fraction is IgG4: functionally deficient for ADCC, complement activation, and Fc-mediated viral clearance, and negatively correlated with the neutralizing titers that standard serology purports to measure. This is not a hypothetical adverse event. It is a documented immunological state in a large population of patients who were told by their clinicians and health authorities that additional boosters would provide additional protection. The adverse event is the gap between what the intervention was represented to deliver and what the immunological evidence shows it actually produced.
The mechanism is local but its adverse consequences are durable. Booster-associated IL-10 does not produce systemic immunosuppression; it produces germinal center tolerization that rewrites the B cell memory pool. IgG4-expressing memory B cells and long-lived plasma cells, once generated, persist for years and continue producing IgG4 at every subsequent antigen encounter. The harm accretes with each booster and does not reverse when vaccination stops. This durability has a direct implication for the standard for action: adverse event surveillance does not require a completed randomized trial to justify concern. The mechanism is established, the magnitude of functional impairment in surrogate assays is published, and the first epidemiological association between the class switch and breakthrough infection is in the peer-reviewed literature. That combination of mechanistic clarity, functional confirmation, and an initial epidemiological signal is precisely the evidentiary pattern that pharmacovigilance systems are designed to act on. The relevant question is not whether causation is proven but whether the risk signal is sufficient to require active surveillance. It is.
The adverse event profile is not uniform across populations. For high-risk patients (the elderly, the immunocompromised, those with significant metabolic or cardiopulmonary comorbidities) the tolerogenic shift toward IgG4 may reduce the most dangerous inflammatory adverse events: cytokine storm, ARDS, and thromboinflammatory sequelae. For these patients, the adverse event calculus may genuinely favor continued boosting with interval discipline and platform optimization. Their immunological adverse event risk from IgG4 accumulation must be weighed against a real and quantified benefit from severe disease prevention.
For healthy children and young adults the calculus is entirely different, and the adverse event burden is not offset by a commensurate benefit. These patients face near-zero individual risk from severe COVID-19. The public health justification for vaccinating them rests on transmission reduction, yet the tolerogenic antibody profile that repeated boosting produces is poorly suited to sterilizing immunity. The adverse events documented in this population (IgG4 switching after two pediatric doses, functional Fc effector impairment, durable memory encoding, and immunological imprinting constraining future humoral adaptability) are being imposed on individuals for whom the clinical benefit is unestablished. Pediatric clinicians should treat this asymmetry as a clinical ethics issue, not merely a policy preference.
The mechanistic pathways driving the adverse event profile are not independent; they are synergistic. Pseudouridine-mediated attenuation of innate inflammatory signaling reduces the pro-inflammatory milieu that would otherwise compete with IgG4 induction. Signal stacking from closely spaced boosters prevents full regulatory cytokine resolution before the next dose. Sustained cGAS-STING activation from residual DNA fragments adds a compounding driver of IL-10 and Treg expansion. Together, these pathways create a positive immune regulatory feedback loop that intensifies with each additional booster. The prolonged germinal center activity uniquely induced by mRNA-LNP platforms (antigen detectable in lymph nodes for up to 60 days, GC reactions persisting at least six months27) provides the sustained substrate for iterative class-switch recombination. The result is that the mRNA-LNP platform, by virtue of the same GC persistence properties that make it immunologically superior for affinity maturation, is also structurally prone to generating progressive IgG4 dominance under conditions of frequent re-dosing. This is not a safety failure unique to a specific manufacturer; it is a platform-level adverse event risk that applies across all pseudouridine-modified mRNA vaccines administered repeatedly.
The regulatory endpoint failure is inseparable from the adverse event surveillance failure. Total anti-spike IgG titer, the primary surrogate used to authorize repeated boosting, is structurally blind to the adverse events this review documents. A fourfold post-booster rise in anti-spike IgG can reflect robust IgG1/IgG3 effector immunity or IgG4 accumulation that hollows out Fc function while preserving the titer. These are clinically non-equivalent outcomes that current assays cannot distinguish and that regulatory frameworks have not required to be distinguished.
The IgG4 class switch was not anticipated when booster schedules were designed, not captured by the endpoints used to authorize them, and was discovered only through post-authorization immunological research that was neither mandated nor standardized. Every clinician relying on post-vaccination serology to counsel a patient about protection should understand that the assay tells them nothing about the subclass composition of that response. In patients who have received four or more boosters, that subclass composition may be the most clinically relevant feature of their humoral immunity, and it is currently invisible to the standard of care.
9. CONCLUSIONS AND POLICY RECOMMENDATIONS
The evidence reviewed here supports the following adverse event–oriented conclusions, clinical action items, and surveillance recommendations:
• Adverse event risk is population-specific and must drive risk-stratified policy. For high-risk patients (elderly, immunocompromised, metabolically vulnerable), the adverse event calculus may favor continued boosting: the documented benefit against severe inflammatory disease is real, and tolerogenic IgG4 properties may reduce harmful sequelae. Boosting should continue in this group but with interval discipline and platform optimization to limit IgG4 accumulation. For healthy children and young adults with negligible severe disease risk, the adverse event burden documented here (durable Fc effector erosion, immunological imprinting, and IgG4 memory encoding after two pediatric doses) is not offset by established clinical benefit. Clinicians advising this group carry an obligation to disclose these trade-offs.
• Booster interval is an adverse event modifiable risk factor. Closely spaced boosters produce signal stacking that amplifies IgG4 induction. Annual minimum spacing, as recommended by Pillai,9 reduces this risk by allowing antigen clearance and regulatory cytokine resolution before re-exposure. Boosters administered more frequently than annually should be treated as carrying a documented adverse immunological cost (progressive IgG4 accumulation and Fc effector erosion) that must be explicitly weighed against the claimed benefit. This trade-off should appear in product labeling and informed consent discussions.
• Accurate mechanistic characterization is prerequisite to effective adverse event surveillance. Booster-associated IL-10 does not produce systemic immunosuppression at documented concentrations; claims that it does are not supported by quantitative data and distort risk-benefit analysis. The genuine adverse event mechanism, germinal center tolerization encoding durable IgG4 memory, is distinct, well-supported, and clinically significant. Conflating these two different claims undermines both clinical credibility and regulatory decision-making.
• IgG subclass assays are the minimum diagnostic tool required to detect the primary adverse event. Total anti-spike IgG is an adverse event–blind surrogate in multiply-boosted patients. IgG subclass analysis must become a standard component of post-vaccination immune assessment in clinical trials, in regulatory submissions for booster authorization, and in routine clinical practice for immunocompromised or oncology patients making booster decisions. Regulatory frameworks should require that applicants for additional booster authorizations demonstrate the intervention does not progressively worsen the IgG1/IgG4 ratio. Clinical laboratories should develop and validate IgG subclass assays suitable for routine diagnostic use; without this infrastructure, the adverse events documented here will remain clinically invisible regardless of how well characterized they become in the research literature.
• Adverse event disclosure requires absolute, not relative, risk metrics. Relative risk reduction values, presented without absolute context, structurally obscure the comparison between benefit magnitude and adverse event risk magnitude. This matters directly for informed consent: a patient who is told a booster reduces their relative infection risk by 40% but is not told the absolute risk reduction is 0.3 percentage points cannot meaningfully weigh that benefit against the adverse immunological consequences documented here. Absolute risk reduction must be the primary efficacy metric in communications to patients, clinicians, and policymakers.
• The adverse event profile differs by the clinical endpoint being pursued. IgG4-dominant immunity reduces severe disease risk (via neutralization, reduced inflammatory pathology) but increases breakthrough infection risk (via ADCC and Fc effector loss). These are not equivalent outcomes and they are not interchangeable in policy justification. A vaccination strategy that is net-beneficial for severe disease prevention may simultaneously be net-harmful for infection prevention in low-risk populations. Policy authorizations, informed consent frameworks, and clinical guidelines must specify which endpoint is being addressed, because the adverse event balance differs materially between them.
• Pediatric booster policy requires formal adverse event re-evaluation with IgG subclass data as a primary outcome. IgG4 class switching has been documented in children aged 5–11 years after only two 10-μg BNT162b2 doses.6 Pediatric guidelines derived by downward extrapolation from adult high-risk data do not account for the fundamentally different adverse event calculus in a population with near-zero severe disease risk. Adverse immunological events that carry acceptable residual weight against substantial mortality benefit in elderly immunocompromised adults carry unacceptable weight in healthy children for whom no comparable mortality benefit exists. Pediatric clinicians must treat the IgG4 switching data as directly relevant to booster counseling for healthy children and must discuss these adverse event risks explicitly with families, not present continued boosting as routine standard of care.
• Prospective adverse event surveillance incorporating IgG subclass assays is the minimum necessary response to the current evidence. The specific priorities are: (a) prospective cohort studies stratifying COVID-19 outcomes by IgG4/IgG1 ratio in multiply-boosted individuals; (b) systematic pharmacovigilance analysis of IgG4-RD case reports temporally associated with repeated mRNA vaccination; (c) oncology registry studies examining outcomes in patients receiving ADCC-dependent therapies concurrent with multiple boosters; (d) immunogenicity studies of co-administered vaccines in multiply-boosted versus singly-vaccinated individuals; (e) pediatric longitudinal studies with IgG subclass outcomes and long-term immune competence follow-up. Post-authorization safety studies of this kind are standard requirements for products with identified adverse event signals. The IgG4 switching data constitutes such a signal. The surveillance infrastructure to characterize it should have been mandated at the time the signal was identified in the peer-reviewed literature.
The adverse events documented in this review are not speculative and are not confined to a single mechanistic claim. Increased breakthrough infection risk has an epidemiological signal. ADCC, complement, and Fc effector function impairments are directly measured in published assays. Durable IgG4 memory encoding is confirmed in longitudinal cohort data. IgG4 class switching in children after two pediatric doses is established. Mechanistically supported pathways (IgG4-RD precipitation, cancer surveillance impairment via ADCC erosion, therapeutic antibody interference, impaired immunity to unrelated pathogens, and immunological imprinting constraining future humoral responses) are grounded in well-characterized immunological biology, and the absence of prospective epidemiological confirmation reflects the absence of surveillance, not the absence of risk.
None of these findings require abandoning mRNA vaccination, which remains effective against severe disease and death in high-risk populations. They do require abandoning the position that repeated boosting is uniformly safe and beneficial across all populations, that total antibody titer is an adequate measure of the immune response it produces, and that post-authorization immunological surveillance is optional. Patients who received multiple boosters under those assumptions, and the clinicians who administered them, deserve an accurate accounting of what the immunological evidence now shows.
DECLARATIONS
Conflicts of Interest: The author declares no conflicts of interest. The author formerly served as the vice chairperson of the CDC ACIP committee, chairperson of the influenza vaccine work group, and member of the COVID work group, but, consequent to a recent court decision in the case of AAP vs HHS, no longer has any affiliation with the CDC or the ACIP. The views expressed are those of the author alone, and do not reflect the opinions of the USG, HHS, CDC, or ACIP.
Funding: No external funding was received for this work.
Ethics Statement: This is a review article. No human subjects research was conducted.
REFERENCES
1. van de Veen W, et al. IgG4 production is confined to human IL-10–producing regulatory B cells that suppress antigen-specific immune responses. J Allergy Clin Immunol. 2013;131(4):1204–1212.
2. Martín Pérez et al. Post-vaccination IgG4 and IgG2 class switch associates with increased risk of SARS-CoV-2 infections. J Infect. 2025;90(4):106473.
3. Class switch toward IgG2 and IgG4 is more pronounced in BNT162b2 compared to mRNA-1273 COVID-19 vaccinees. Int J Infect Dis. 2025;159:107990.
4. Ravichandran S [Repeated COVID-19 mRNA vaccination results in IgG4 class switching and decreased NK cell activation by S1-specific antibodies in older adults]. Immun Ageing. 2024;21(1):63.
5. Class switch towards spike protein-specific IgG4 antibodies after SARS-CoV-2 mRNA vaccination depends on prior infection history. Sci Rep. 2023;13:13166.
6. Kobbe R, et al. Delayed induction of noninflammatory SARS-CoV-2 spike-specific IgG4 antibodies detected 1 year after BNT162b2 vaccination in children. Pediatr Infect Dis J. 2024;43(12):1200–1203. doi:10.1097/INF.0000000000004516.
7. Irrgang P, et al. Class switch toward noninflammatory, spike-specific IgG4 antibodies after repeated SARS-CoV-2 mRNA vaccination. Sci Immunol. 2023;8(79):eadd2032. doi:10.1126/sciimmunol.add2032.
8. Kalkeri R, et al. Priming platform determines IgG4 class switching and Fc effector function after COVID-19 booster vaccination: post-hoc immunological analysis of four clinical trials comparing mRNA and protein subunit platforms. J Infect. 2025. doi:10.1016/j.jinf.2025.106473. [Funded by Novavax; majority of authors are Novavax employees.]
9. Pillai S. Is it bad, is it good, or is IgG4 just misunderstood? Sci Immunol. 2023;8(81):eadg7327. doi:10.1126/sciimmunol.adg7327.
10. Wilks SH, Moller R, Andersen KG, et al. SARS-CoV-2 evolution on a dynamic immune landscape. Nature. 2025.
11. Weisblum Y, Schmidt F, Zhang F, et al. Escape from neutralizing antibodies by SARS-CoV-2 spike protein variants. eLife. 2020;9:e61312.
12. Ellerhorst JA, et al. Comparison of serum interleukin-10 (IL-10) levels between normal volunteers and patients with advanced melanoma. Melanoma Res. 2001;11(3):247–252.
13. Sarris AH, et al. Interleukin-10 levels are often elevated in serum of adults with Hodgkin’s disease and are associated with inferior failure-free survival. Ann Oncol. 1999;10(4):433–440.
14. Rosati M, et al. Cytokine signature following COVID-19 mRNA booster vaccination in previously vaccinated adults. Front Immunol. 2023;14:1292568. doi:10.3389/fimmu.2023.1292568.
15. Longitudinal cytokine study in frail elderly following COVID-19 mRNA booster vaccination. Immun Ageing. 2024;21:44.
16. Gao Q, et al. High level of interleukin-10 in serum predicts poor prognosis in multiple myeloma. Br J Cancer. 2016;114(4):463–469.
17. Nakamura Y, et al. High levels of baseline serum IL-10 are associated with reduced clinical benefit from first-line immune checkpoint inhibitor therapy in advanced renal cell carcinoma. Front Oncol. 2023;13:1134988.
18. Heeschen C, et al. Serum level of the antiinflammatory cytokine interleukin-10 is an important prognostic determinant in patients with acute coronary syndromes. Circulation. 2003;107(16):2109–2114.
19. Wojciechowski W, et al. Immune regulation and cytotoxic T cell activation of IL-10 agonists: preclinical and clinical experience. Front Immunol. 2020;10:2922.
20. Ouyang W, O’Garra A. IL-10 family cytokines IL-10 and IL-22: from basic science to clinical translation. Immunity. 2019;50(4):871–891.
21. Akhtar M, et al. Appearance of tolerance-induction and non-inflammatory SARS-CoV-2 spike-specific IgG4 antibodies after COVID-19 booster vaccinations. Front Immunol. 2023;14:1309997.
22. Massanella M, et al. IL-10 indirectly downregulates IL-4–induced IgE production by human B cells. ImmunoHorizons. 2018;2(11):398–408.
23. Boonpiyathad T, et al. The role of IgG4 in the fine tuning of tolerance in IgE-mediated allergy and cancer. Int J Mol Sci. 2020;21(14):5017.
24. Jutel M, et al. IL-10 and TGF-β cooperate in the regulatory T cell response to mucosal allergens in normal immunity and specific immunotherapy. Eur J Immunol. 2003;33(5):1205–1214.
25. Tsitsiklis A, et al. IL-10–producing Tfh cells accumulate with age and link inflammation with age-related immune suppression. Sci Adv. 2020;6(33):eabb0806.
26. IL-10 effects on STAT3, Treg, and NK suppression in tumor microenvironment. [Tumor immunology literature; specific citation per manuscript records.]
27. Kim W. Germinal center response to mRNA vaccination and impact of immunological imprinting on subsequent vaccination. Immune Netw. 2024;24(4):e28. doi:10.4110/in.2024.24.e28.


Dr Malone, I want to thank you for the intellectual master class explanation. I am a pathologist who retired with 35 yrs practicing anatomic/clinical/neuropathology rather than take the “vax”. Subsequently I have seen a number of my former colleagues experience the now well documented myocarditis, strokes, autoimmune diseases and death. I have subscribed for over a year and forward your vaccine and immunology articles to former colleagues and new docs in my former private practice group.
Thank you once again
Thank you for further sharing this!
I personally consider it beyond regrettable you were prevented from presenting it in a forum where the issues you cover could be considered and would potentionally actionable.
I would note, as a member of the High Risk group and reviewing the risks, I find it difficult to agree to the vaccination.
You are appreciated! IMO the Court causes great harm to we the people!