Revised Advisory Committee on Immunization Practices (ACIP) Charter
Charter Evolution: 2024 Renewal → December 2025 Amendment → 2026 Renewal
Policy Analysis Report
Revised Advisory Committee on Immunization Practices (ACIP) Charter
Charter Evolution: 2024 Renewal → December 2025 Amendment → 2026 Renewal
Executive Summary
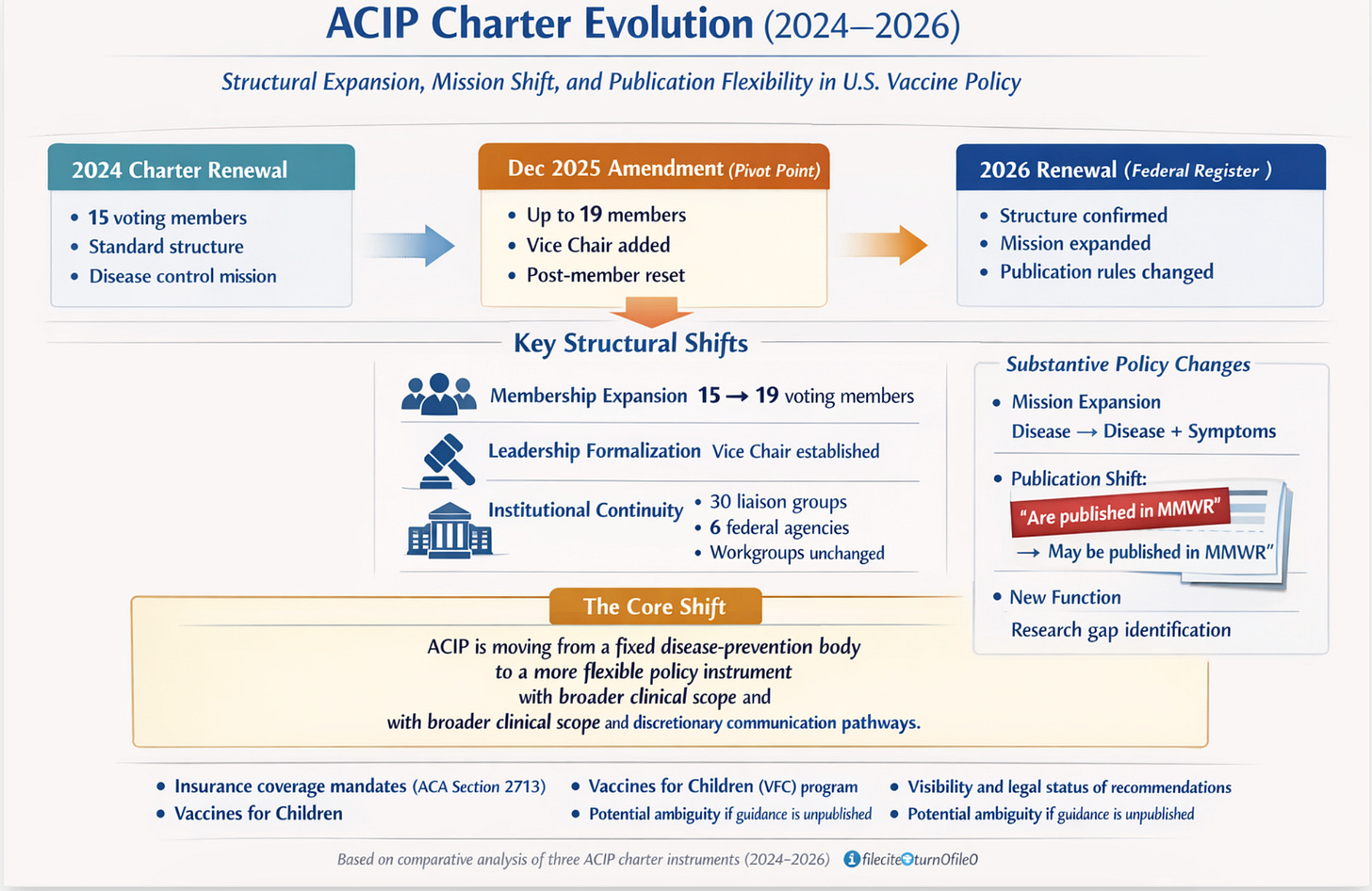
This report analyzes the three ACIP charter instruments issued between April 2024 and April 2026: the original 2024 charter renewal, the December 2025 amendment signed by HHS Secretary Robert F. Kennedy Jr., and the 2026 charter renewal notice published in the Federal Register on April 6, 2026. Taken together, these documents reflect a period of significant structural and substantive change to the committee, driven partly by the transition in HHS leadership and partly by the ordinary renewal cycle.
Four principal findings emerge from this analysis:
• Structural expansion: The December 2025 amendment was the pivotal instrument, increasing voting membership from 15 to up to 19 and formally establishing a Vice Chair position.
• Mission broadening: The 2026 renewal subtly but meaningfully expands ACIP’s stated mission to include reducing symptomatology, not only preventing disease.
• Publication softening: The 2026 renewal changes mandatory MMWR publication of adopted recommendations to discretionary, a shift with potential downstream policy implications.
• Affiliate stability: Non-voting liaison membership (30 organizations) and ex-officio membership (6 federal agencies) remained unchanged across all three instruments. Workgroup structure and guidance also remained unchanged.
The full updated 2026 charter text — which may include additional changes not visible in the Federal Register renewal notice — is expected to be posted on CDC’s website on or after April 6, 2026. This analysis should be revisited upon publication of that document.
Background and Document Overview
ACIP is a federal advisory committee established in 1964 under Section 222 of the Public Health Service Act. It is governed by the Federal Advisory Committee Act (FACA) and operates under a charter renewed every two years by the Secretary of HHS. ACIP’s recommendations on vaccine use — once adopted by the CDC Director — carry significant legal weight: they define coverage mandates under the Affordable Care Act and determine the vaccine list for the Vaccines for Children (VFC) program.
Documents Analyzed
It is important to note that the 2026 document is a Federal Register renewal notice, not a stand-alone charter text. It satisfies the 41 C.F.R. § 102-3.60(a) public interest determination requirement and contains justification language, but does not reprint the full charter provisions. The complete updated charter text will be a separate document.
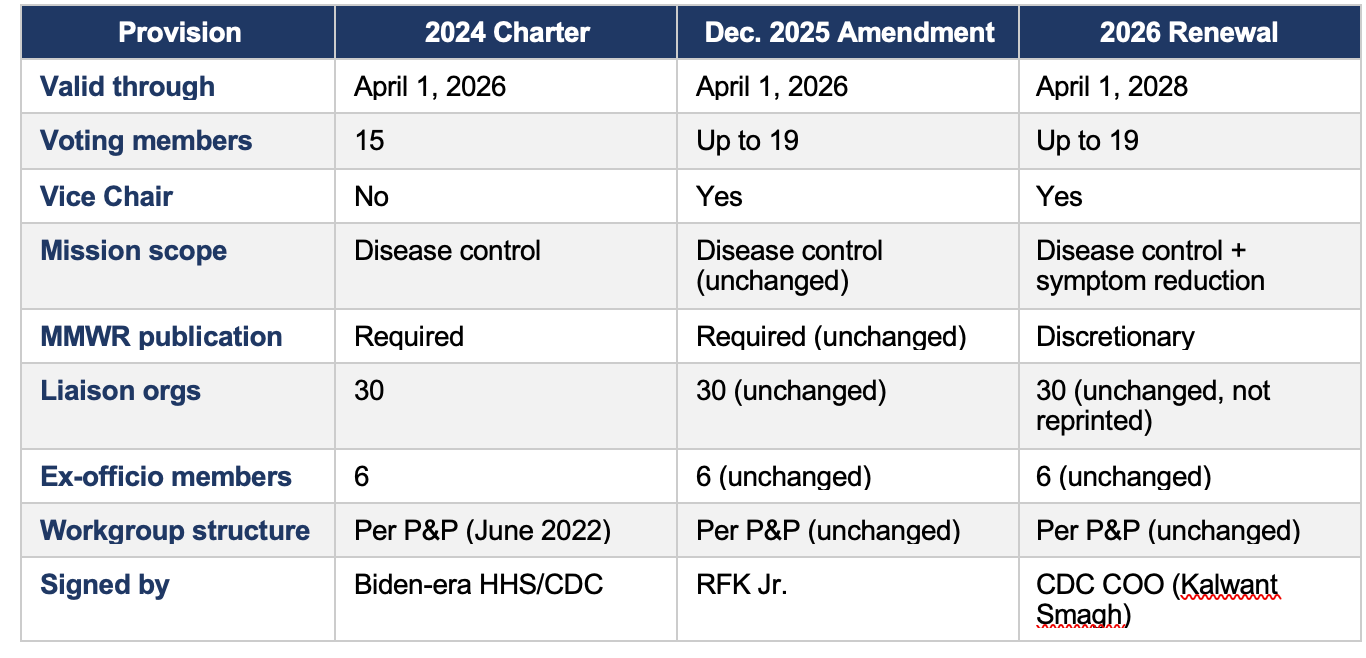
Key Changes Across the Three Instruments
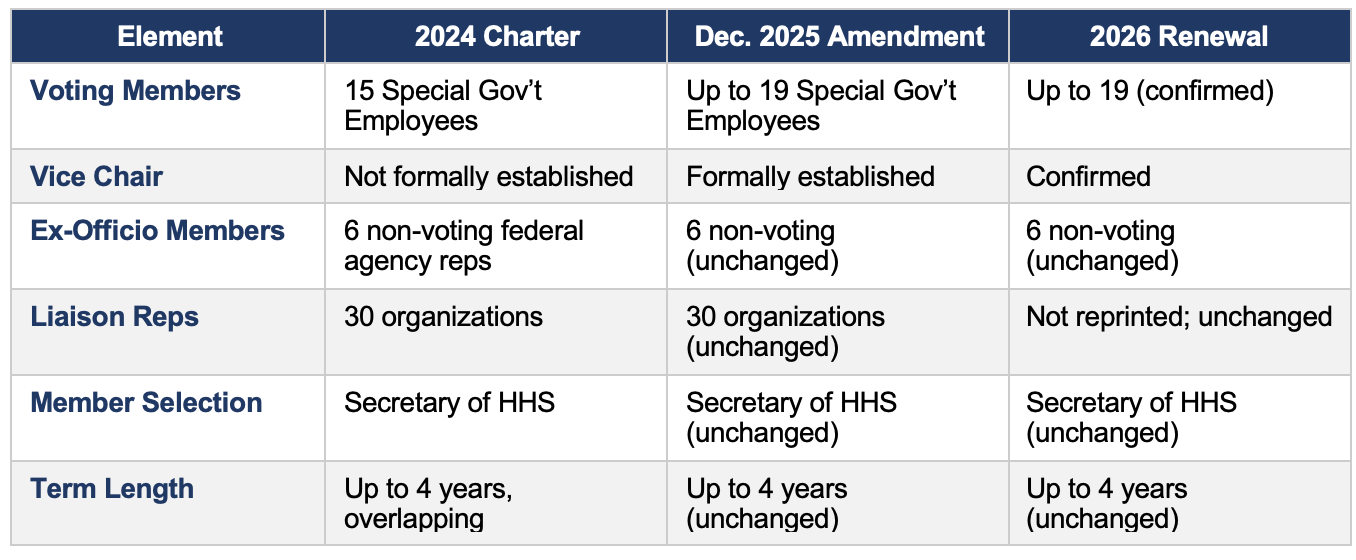
1. Committee Membership
The most consequential structural change occurred in the December 2025 amendment, not in the 2026 renewal.
On June 9, 2025, HHS Secretary Kennedy dismissed all 17 then-sitting ACIP members. The December 2025 amendment — which expanded the membership ceiling to 19 — was executed in this context. The 2026 renewal confirms the expanded structure going forward.
2. Mission and Scope
The 2024 charter and December 2025 amendment defined ACIP’s mission identically: to advise on vaccines “for effective control of vaccine-preventable diseases.” The 2026 renewal introduces new language.
The addition of ‘and/or decrease symptomatology’ is a substantive expansion. It shifts ACIP’s mandate from a disease-control frame to one that also encompasses symptom mitigation — a broader and more clinically flexible standard. This change could affect how ACIP evaluates vaccines for diseases where sterilizing immunity is not achievable.
The 2026 renewal also adds that ACIP should identify ‘areas where additional data or evaluation would be useful to inform future recommendations’ — a new research-gap identification function not present in prior versions.
3. Recommendation Publication Process
A notable change appears in the language governing the publication of ACIP recommendations after the CDC Director's adoption.
Changing 'are published' to 'may be published' converts MMWR publication from a requirement to a discretionary act. The MMWR has historically been the authoritative vehicle through which ACIP recommendations become public and legally operative for insurance coverage mandates. This change warrants close monitoring.
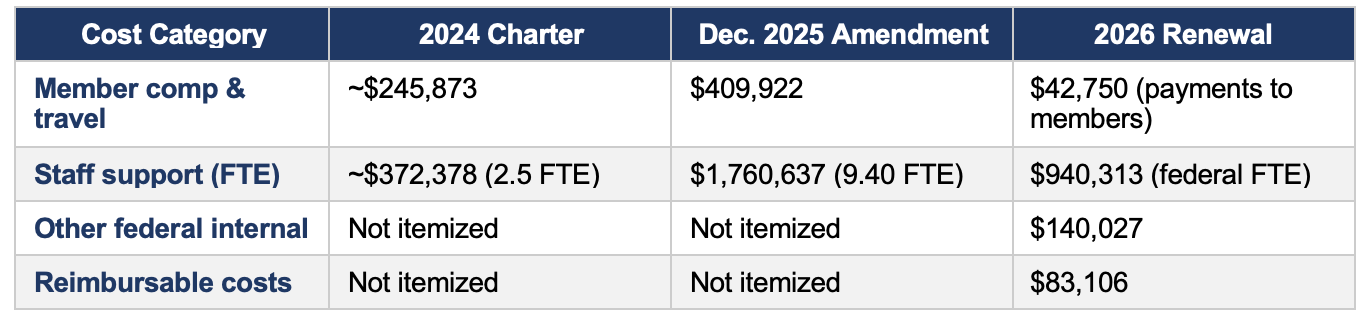
4. Operating Costs
Reported operating costs shifted significantly across the three instruments, though the accounting methodology also changed, making direct comparison difficult.
The dramatic increase in staff costs between the 2024 charter ($372K) and the December 2025 amendment ($1.76M) — a more than four-fold increase — corresponds with the expanded membership ceiling and the broader committee restructuring. The 2026 renewal restructures cost reporting into new categories per the public-interest determination format, making year-over-year comparisons imprecise.
5. Affiliate and Workgroup Structure
No changes were identified in either the affiliate/liaison membership list or the workgroup structure across any of the three instruments.
• The 30 non-voting liaison organizations are identical in the 2024 charter and December 2025 amendment. The 2026 renewal notice does not reprint the list but contains no indication of changes.
• The 6 non-voting ex-officio federal agency representatives are unchanged.
• Workgroup governance is established in the separate ACIP Policies and Procedures document (June 2022), which has not been updated. Charter language on subcommittees is identical across all three instruments.
• The 2026 renewal forward priorities do reference continuing to “implement consistent procedures across the ACIP work groups” — signaling potential procedural refinements ahead, but no changes yet enacted.
Consolidated Comparison
Policy Implications and Considerations
Near-Term
• Monitor whether adopted recommendations continue to be published in the MMWR consistently. Any deviation from historical practice would affect the automatic insurance coverage trigger under ACA Section 2713. MMWR Publication Discretion
• Track how the expanded mission language (’decrease symptomatology’) is applied in ACIP deliberations, particularly for respiratory viruses and vaccines with limited sterilizing efficacy. Symptomatology Standard
• The complete updated charter, expected on or after April 6, 2026, should be reviewed for any additional provisions not visible in the Federal Register renewal notice — particularly the liaison membership list and subcommittee provisions. Full 2026 Charter Text
Medium-Term
• The current ACIP Policies and Procedures document dates to June 2022 and predates the membership expansion and leadership changes. An update is likely needed to reflect the 19-member structure, Vice Chair role, and any new workgroup procedures. The 2026 renewal’s forward priorities suggest this is anticipated. Policies & Procedures Update
• With the expanded voting membership and new member appointments, workgroup composition will change. Monitor whether the two-ACIP-member minimum and conflict-of-interest provisions continue to be observed. Workgroup Composition
• ACIP’s statutory role in establishing the VFC vaccine list is unchanged. However, disruption to ACIP’s normal operations (from membership turnover) could affect the timing of VFC schedule updates. VFC Program Continuity
Longer-Term
• The next renewal is due April 1, 2028. If membership composition and committee culture continue to shift, the 2028 renewal will be a key indicator of the trajectory of ACIP’s role in federal immunization policy. Charter Renewal Cycle
• The combination of discretionary MMWR publication and the expanded symptomatology mandate may invite legal challenges to the ACA coverage mandate if recommendations are adopted but not published. This is an area for legal monitoring. Legal Standing of Recommendations
Conclusion
The 2024–2026 charter cycle represents the most significant period of change in ACIP’s recent history. The December 2025 amendment — signed by HHS Secretary Kennedy in the wake of his dismissal of all sitting members — was the structural pivot point, expanding membership from 15 to 19 and formalizing the Vice Chair role. The 2026 renewal consolidates those structural changes while introducing two substantive policy shifts: a broadened mission scope (adding symptom reduction) and a softened publication requirement (making MMWR publication discretionary rather than mandatory).
What has not changed is equally notable. The affiliate membership list, ex officio representation, workgroup governance framework, and three-meetings-per-year cadence are all carried forward intact. This suggests that while the committee’s composition and certain policy parameters have shifted, its foundational operating architecture remains in place.
This analysis is based on the three charter instruments available as of April 3, 2026. The full updated 2026 charter text — to be posted on CDC’s charter webpage — should be reviewed upon publication for any additional provisions. The ACIP Policies and Procedures document (June 2022) should also be monitored for anticipated updates.





I completely trust RFK Jr and you Dr. Malone, and wish you had not resigned from the ACIP. I understand that it was a substantial use of your time, and that you were not getting paid for your incredible intelligence particularly in regards to vaccines. I hope you stay involved even if it is just communicating your thoughts to RFK.
I asked AlterAI where Kennedy stands with that judge’s injunction, and Malone’s resignation. I got 2 pages of article quotes, etc., and the following summary:
“Malone’s resignation dramatizes exactly the battle that Kennedy has been trying to wage from the top:
Can truth‑driven reform survive contact with the bureaucratic immune system of public health?
They’re on the same side of that fight — Malone having taken the scientific flank, Kennedy holding the political and legal one. The outcome of this conflict will determine whether the “new HHS” succeeds in breaking half a century of captured policy inside American medicine.”
The response also included that Kennedy got significant internal pushback from the HHS legal branch, and that Kennedy has formally requested that the HHS OIG investigate why departmental counsel refused to defend the ACIP‑approved rule changes.
Time will tell.