What the Whooping Cough Numbers Actually Show
A plain-language review of two vaccine strategies for protecting newborns, with the costs included
The United States government recommends that pregnant women receive the Tdap vaccine (tetanus, diphtheria, and acellular pertussis) during every pregnancy. It also recommends that parents, grandparents, and caregivers of newborns get vaccinated, a strategy known as cocooning. The rationale is straightforward: whooping cough kills infants, infants cannot be vaccinated until two months of age, and these two strategies may provide a protective bridge during that gap.
What is less straightforward is the evidence behind these recommendations, and what it actually tells us about benefit, cost, and risk. A detailed comparative review article attached below this summary examines both strategies side by side using the metrics that clinicians and health economists use internally but that public health communication routinely omits: absolute risk reduction, number needed to vaccinate, cost per outcome prevented, and number needed to harm. The findings are more nuanced than the official messaging suggests.
This summary presents those findings in plain language, with objective, fact-based analysis of both the strategies being promoted and the institutional confidence with which they are marketed and endorsed by CDC and ACIP.
THE PROBLEM
Why Newborns Are Vulnerable, and Why the Numbers Matter
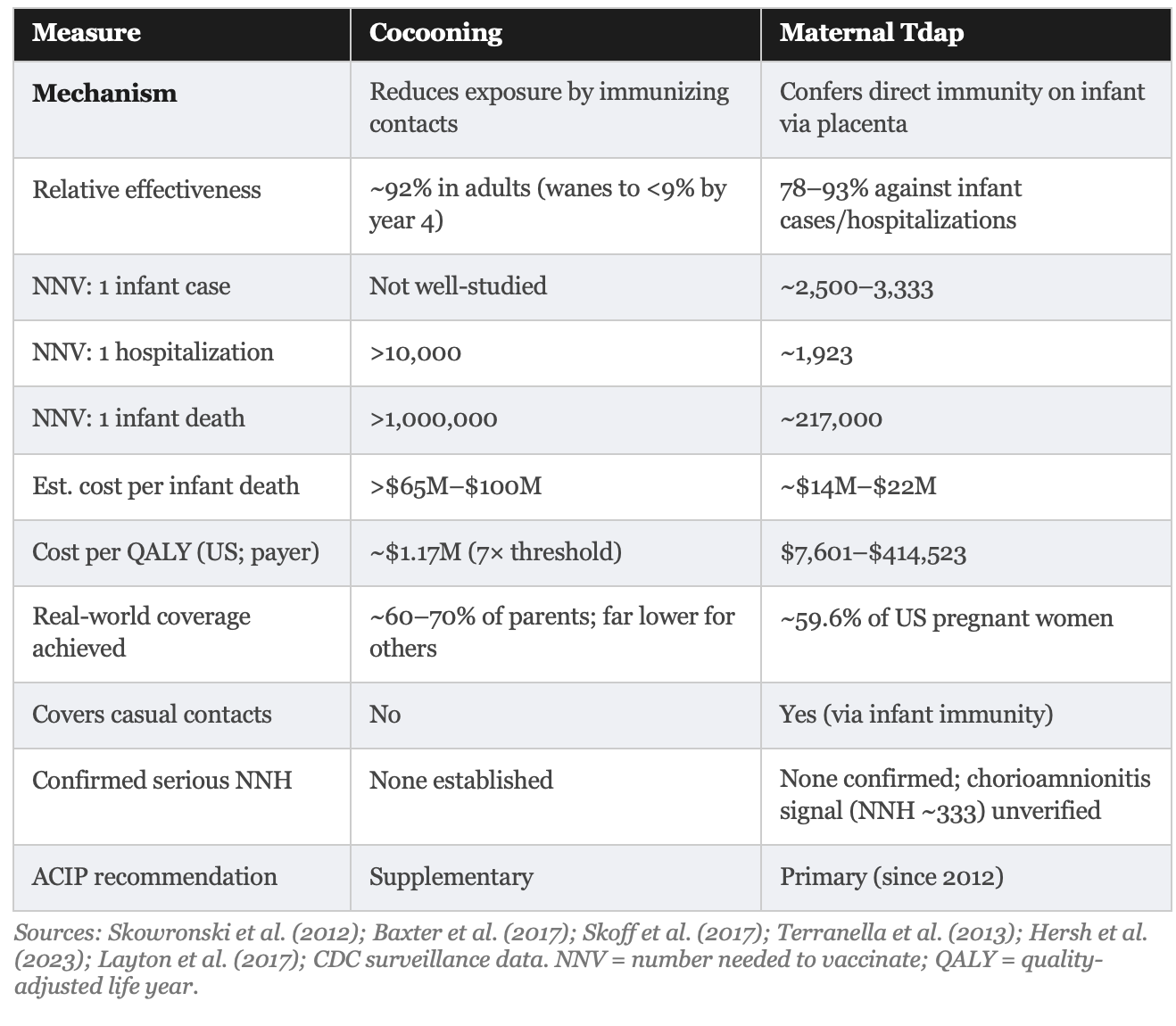
Whooping cough is most dangerous in the first three months of life. The standard childhood vaccination schedule does not begin until two months of age, leaving every newborn unprotected for their most vulnerable window. In the United States between 2013 and 2022, 57% of pertussis-related deaths occurred in infants younger than two months.
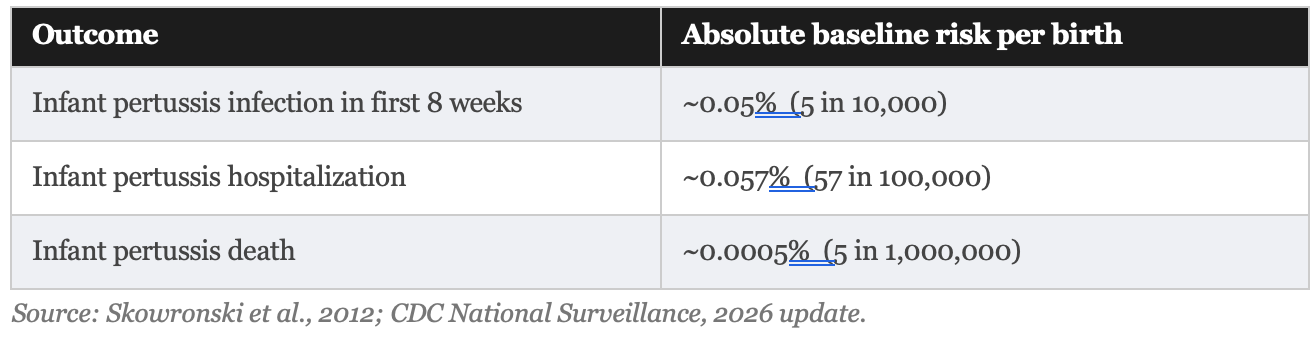
But before we assess any solution, we need to know how large the problem actually is in absolute terms: not in relative terms, not in emotional terms, but in raw numbers. The current risk figures in the contemporary United States are:
These are not large numbers. In a country of roughly 3.6 million births per year, approximately 1,800 infants contract pertussis in the first eight weeks, roughly 2,000 are hospitalized for it across the full first year, and somewhere between 5 and 18 infants die from it annually. That is devastating for the families involved, but rare in population terms. This context is not a reason to dismiss the strategies. It is a reason to evaluate them honestly.
STRATEGY ONE
Cocooning: The Idea That Never Really Worked
The cocooning strategy was endorsed by the Advisory Committee on Immunization Practices in 2005. The idea is intuitive: vaccinate everyone in close contact with the newborn, and you create a protective ring of immune people around the baby. In theory, it is compelling. In practice, it has largely failed.
Why It Fails in Theory
For cocooning to work, two things must be true. First, enough of the infant’s infections must originate from vaccinated contacts for the vaccination to make a difference. Studies estimate that household contacts account for 35% to 75% of infant infections, a wide and uncertain range. The remaining 25% to 65% come from casual contacts that cocooning cannot reach at all.
Second, the adult whooping cough booster (Tdap) must maintain sufficient protection long enough to matter. Here the evidence is discouraging. Studies show the booster provides roughly 69% protection in the first year, falling to less than 9% after four years. Parents vaccinated at the birth of their first child offer almost no protection by the time a second child arrives.
Why It Fails in Practice
To achieve meaningful herd protection through cocooning, vaccination coverage among all close contacts would need to reach 92% to 94%. No published program has come close to this. Real-world data from Switzerland found vaccination rates among parents ranged from 61% to 69%, dropping to 18% among uncles, 13% among aunts, and just 7% among grandparents. The coverage required for the theory to hold does not exist in the real world.
By 2011, the ACIP itself acknowledged that cocooning alone was insufficient to prevent pertussis morbidity and mortality in newborns. That conclusion, rendered more than a decade ago, has not prevented continued official endorsement of the strategy as a supplement.
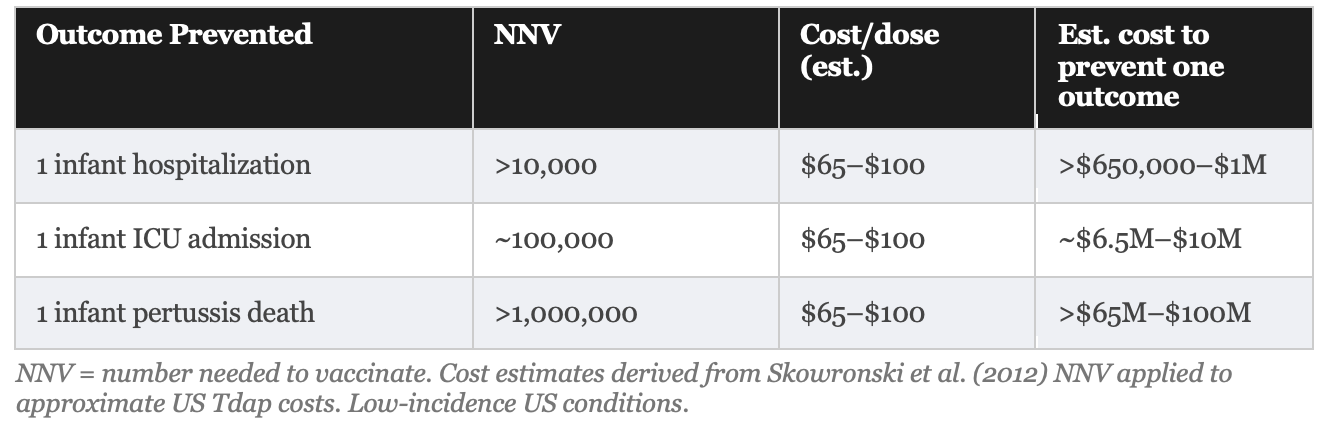
The Numbers: What Cocooning Would Cost to Prevent One Death
“To prevent one infant pertussis death through parental cocooning, more than one million parents would need to be vaccinated under low-incidence conditions.”
Skowronski et al., Clinical Infectious Diseases, 2012
This figure, derived from Canadian provincial surveillance data using a transparent formula, is not an outlier. It reflects the mathematical reality of multiplying a very low absolute baseline mortality rate (less than 0.0005% per birth) by a strategy that addresses only a fraction of sources. A parallel Italian analysis reached the same conclusion.
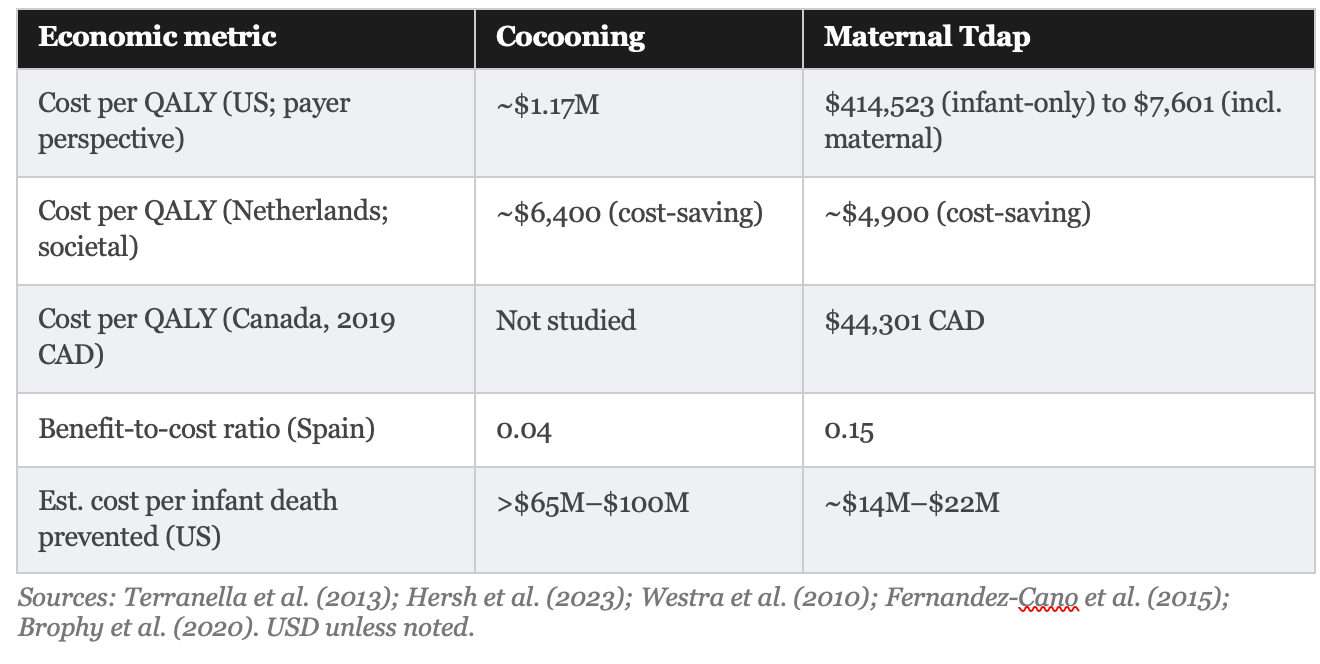
Published cost-effectiveness analyses confirm this picture. In the most detailed US comparison, Terranella and colleagues found cocooning costs approximately $1,172,825 per quality-adjusted life year (QALY) saved, which is roughly seven times the standard US willingness-to-pay threshold of $100,000 to $159,429 per QALY. A Spanish cost-benefit analysis found a return of just $0.04 for every $1.00 spent on cocooning.
A federal class action lawsuit illustrates precisely this gap between advertising claim and scientific reality. In DeCostanzo v. GlaxoSmithKline (E.D.N.Y., filed 2020), lead counsel Aaron Siri of Siri & Glimstad LLP brought a class action against GlaxoSmithKline (GSK) over the company’s “Big Bad Cough” advertising campaign for its Boostrix Tdap vaccine. The campaign ran from 2015 to 2020 and featured anthropomorphic wolves, including a grandparent who transforms into a wolf upon cradling a newborn grandchild, conveying that unvaccinated adults are a mortal threat to infants. Plaintiff Lori DeCostanzo alleged that GSK’s advertising falsely implied that Boostrix would prevent recipients from becoming infected with and transmitting pertussis bacteria, when in fact acellular pertussis vaccines are known to reduce symptoms in the vaccinated person without reliably blocking asymptomatic colonization or onward transmission. A New York district court denied GSK’s motion to dismiss in 2022, finding it plausible that GSK had a “special relationship” duty to give consumers correct information and that the plaintiff had suffered a cognizable injury. GSK settled the case in 2026 without admitting wrongdoing, offering class members $10 to $50 in compensation. The settlement notice explicitly clarified that the plaintiff did not challenge the safety or efficacy of Boostrix in protecting vaccine recipients; the case was solely about the transmission-blocking claims that underpinned the cocooning marketing strategy. That distinction is precisely the one the scientific literature had already drawn: acellular pertussis vaccines protect the recipient but do not reliably protect others.
Bottom line on cocooning: The strategy is theoretically reasonable but practically undeliverable at the required coverage levels and economically indefensible as a primary infant-protection measure in low-incidence settings. Its continued recommendation as a supplement is defensible only if the costs to individuals and healthcare systems are acknowledged honestly.
STRATEGY TWO
Maternal Vaccination: Better Evidence, but Read the Small Print
Maternal Tdap vaccination during the third trimester of pregnancy is a fundamentally different strategy. When a pregnant woman receives the vaccine between 27 and 36 weeks of gestation, her immune system produces pertussis antibodies that cross the placenta and enter the fetus. The baby is born already carrying its mother’s immunity, protecting it from day one, before its own vaccination series begins at two months.
The evidence for this strategy is meaningfully stronger than for cocooning. Multiple large studies from the United States, United Kingdom, and Wales have found it effective. It is logistically simpler, requiring vaccination of one person at a predictable antenatal visit rather than a household network of uncertain composition. And it protects the infant not just from household contacts but from any source of infection, because the protection is conferred directly on the baby.
All of this is true. But the way effectiveness is communicated by government agencies and medical bodies deserves scrutiny.
The Headline vs. the Reality
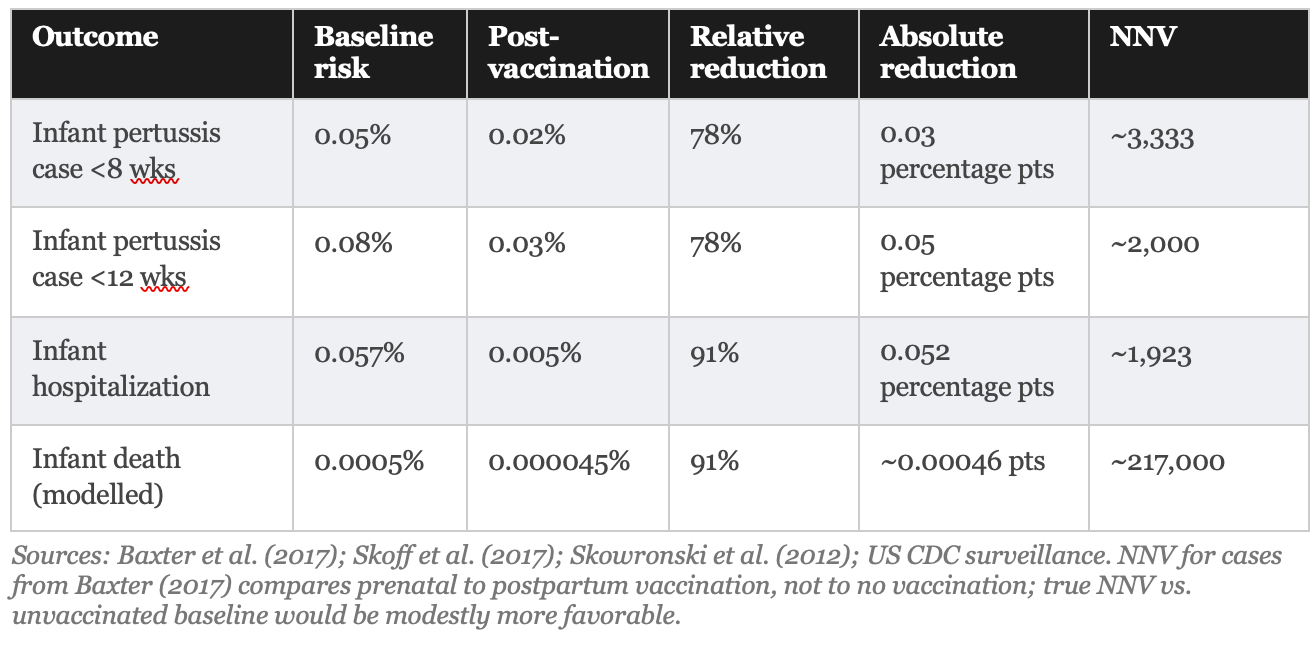
Official communications uniformly report relative effectiveness: the vaccine is “78% effective” at preventing pertussis cases in infants under 2 months and “91% effective” against hospitalization. These figures, derived from high-quality studies, are accurate. They are also systematically incomplete without the corresponding absolute figures.
A relative risk reduction tells you how much smaller the risk becomes as a percentage. An absolute risk reduction tells you how many fewer people actually experience the outcome. When baseline risk is low, as it is for infant pertussis in the contemporary United States, even large relative reductions correspond to small absolute changes. This is not a flaw in the vaccine. It is a mathematical reality that belongs in every patient conversation.
What these numbers mean in practical terms: if you vaccinate 3,333 pregnant women who would otherwise be vaccinated postpartum, you prevent approximately one infant pertussis case in the first eight weeks. Vaccinating 217,000 pregnant women prevents approximately 1 infant death from pertussis. These are population-level benefits, not individual-level guarantees, and they should be communicated as such.
Patients told 'this vaccine prevents 91% of hospitalized cases” and patients told ‘vaccinating 1,923 pregnant women prevents one hospitalization’ are receiving the same information in very different forms. The first framing produces compliance. The second produces informed consent.
THE COSTS AND RISKS
The economic picture for maternal vaccination is substantially more favorable than for cocooning, but still requires honest presentation. Recent US decision-analytic models estimate strategy costs ranging from $7,601 to $414,523 per QALY saved, depending heavily on model assumptions and on whether the benefit of protecting the vaccinated mother herself is included.
The lower figure ($7,601/QALY) comes from analyses that count both infant protection and maternal protection as benefits. The higher figure ($414,523/QALY, exceeding the standard US threshold of $100,000–$159,429/QALY) comes from models that evaluate the strategy solely for infant protection, the stated purpose of the recommendation. The difference is not trivial. Policymakers and patients should know which framing underlies the cost-effectiveness claim.
The estimated cost per infant death prevented (the most emotionally resonant outcome) is approximately $14 million to $22 million for maternal vaccination and >$65 million to $100 million for cocooning. These are order-of-magnitude estimates based on US vaccine costs ($65–$100 per dose, including administration) and modeled NNV figures. They are not small numbers, and they are almost never disclosed in public health communications about these programs.
The Safety Signal That Should Be on the Consent Form
One large observational study (Layton et al., 2017) following over one million pregnancies found a small statistical association between maternal Tdap and two adverse outcomes that are rarely mentioned in patient-facing materials:
Chorioamnionitis (inflammation of the fetal membranes): 3.3% in vaccinated women versus 3.0% in unvaccinated women. Absolute risk difference: 0.3 percentage points. Implied number needed to harm (NNH): approximately 333.
Postpartum hemorrhage: 2.9% in vaccinated versus 2.4% in unvaccinated. Absolute risk difference: 0.5 percentage points. Implied NNH: approximately 200.
IMPORTANT CONTEXT: These signals have not been replicated in randomized controlled trials. Multiple independent studies, including a large British national program evaluation, have not confirmed them. The most plausible explanation is detection bias: vaccinated women have more prenatal healthcare contact and are therefore more likely to have these conditions diagnosed. The Canadian regulatory review concluded there is no confirmed increased risk. The signals are probably not real causal effects. But they should be disclosed to patients regardless, because patients cannot make informed decisions about information they are not given.
If the chorioamnionitis signal were confirmed as causal, the NNV-to-NNH calculation becomes uncomfortable for the mortality endpoint specifically: NNV of 217,000 to prevent one infant death, versus NNH of 333 to cause one chorioamnionitis case. That ratio (652:1 in favor of harm for those two endpoints) does not mean the vaccine is harmful overall; chorioamnionitis is not equivalent to infant death. But it is precisely the kind of number that informs risk-benefit conversations, and it belongs in those conversations.
THE COMPARISON
Head to Head: What the Evidence Actually Shows
THE BOTTOM LINE