What They Don't Tell You About the Shingles Vaccine
The headline number is 97% effective. The number that actually matters to you is closer to 3%. Both are true. Here's what's going on — and why the distinction matters.
THE DISEASE
First, shingles is genuinely unpleasant
To fairly evaluate any vaccine, you have to start with the disease it prevents. Shingles is caused by the same virus that gave you chickenpox as a child, it never fully leaves your body. It retreats into your nervous system and can reactivate decades later, typically producing a painful, blistering rash that appears in a band on one side of your torso, face, or limbs.
For most people, the rash resolves in two to four weeks. But for a significant minority, roughly one in five people who get shingles, rising to nearly one in three among adults over 60, the pain doesn’t stop when the rash heals. This complication is called postherpetic neuralgia, or PHN. It’s a chronic nerve pain that can persist for months, years, or sometimes permanently, and is described by those who have it as burning, electric, and constant. It is associated with depression, sleep disruption, and social withdrawal.
Most cases of shingles are mild to moderate and resolve within a few weeks, with 10–20% becoming “severe” in a meaningful clinical sense.
I have personally had shingles fifteen plus years ago, an unusual and rather nasty variant called intra-abdominal shingles. Inside your abdomen. I did not have medical insurance, and so I delayed treatment until I could not stand the pain. As a consequence, I developed a particularly nasty postherpetic neuralgia- a paralytic polio-like syndrome that caused a partial paralysis of the left side of my abdomen. Suffice to say, this created problems as an advanced equestrian. I quite literally lost my core. That said, I personally consider that a “natural” booster, and have never accepted the shingles vaccine. My body, my choice.
About 10–18% of shingles cases progress to PHN, which is the main long-term issue. There is no “cure” for PHN, as no drug will cure it. The treatments available are nerve-calming drugs, like gabapentin, antidepressants used as pain medications, and lidocaine patches, which help some people, but roughly 40 to 50 percent of PHN patients never achieve adequate pain relief from any available therapy. BTW - stating that there is “no cure” is medically acceptable language, but it lacks nuance. Most people do improve and even achieve complete recovery, gradually over time. But there is no treatment that will “cure” PHN - drugs will only help with the symptoms, so this is a significant nuance.
This matters for understanding the vaccine because preventing shingles also means preventing any chance of developing PHN. But how effective is the vaccine at actually doing that for any individual?
THE NUMBERS
97% effective. What does that actually mean?
The figure comes from two large clinical trials called ZOE-50 and ZOE-70, which together enrolled about 30,000 adults across 18 countries. Participants were randomly assigned to receive either Shingrix or a saline injection, then followed for roughly three to four years to see who developed shingles.
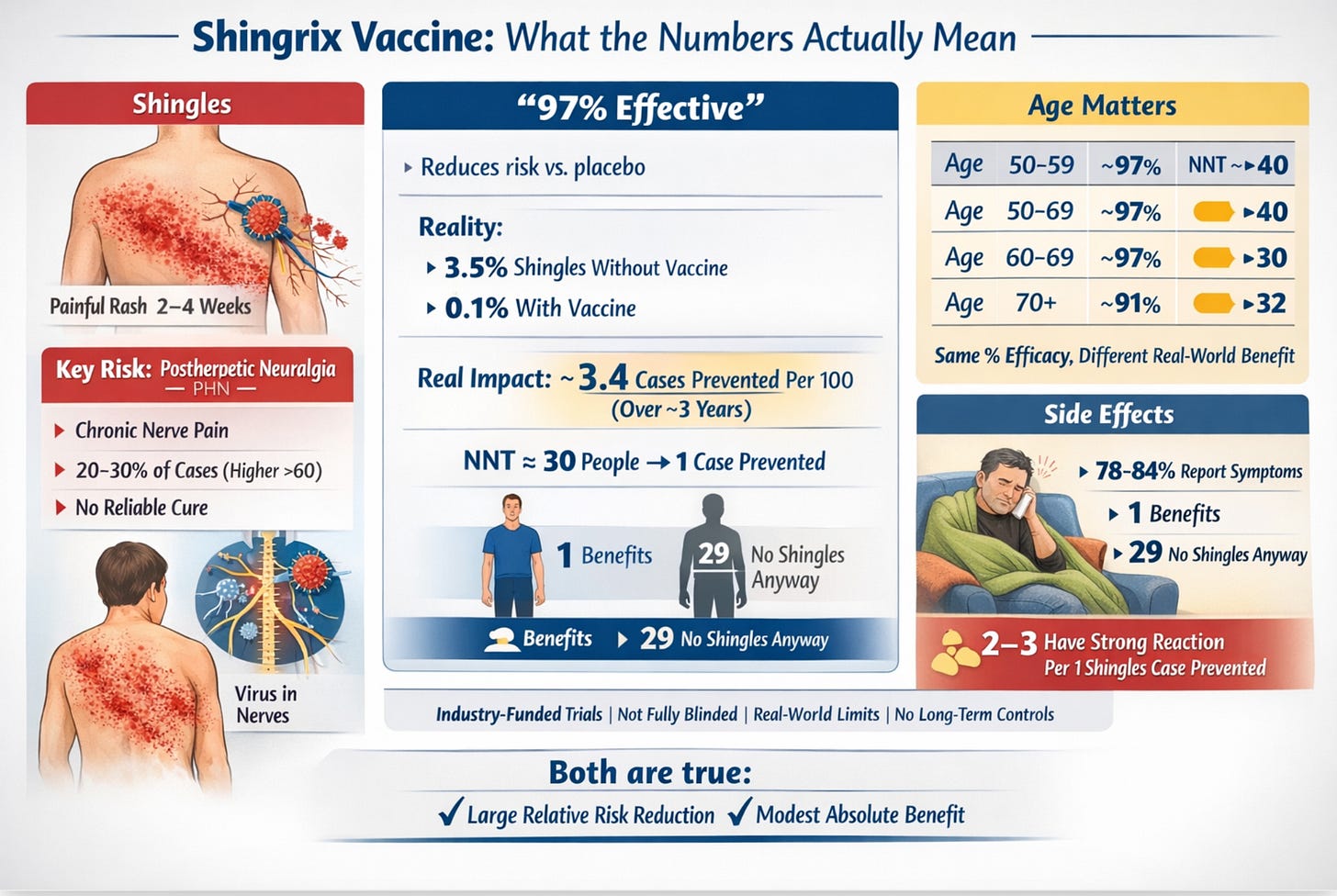
Here are the actual results for adults aged 60 to 69, the age group with the highest shingles rates:
Source: ZOE-50 trial (Lal et al., N Engl J Med, 2015). Modified vaccinated cohort, mean follow-up 3.2 years.
Now you can see both numbers at once. The vaccine group’s rate was about 3% of the placebo group’s rate; that’s where “97% effective” comes from. It’s the ratio. It’s a relative measure.
But look at the placebo column. Only 3.5 out of 100 unvaccinated people in this age group developed shingles over roughly three years. That means 96.5 out of 100 would not have gotten shingles anyway, with or without the vaccine.
“97% effective” means the vaccine reduced the rate among those who would have gotten shingles. It does not mean 97 in 100 vaccinated people are protected from something that would otherwise have happened to them.
The absolute benefit, the actual reduction in cases attributable to vaccination, was about 3.4 cases prevented per 100 people vaccinated over three years. That translates to a “number needed to treat” (NNT) of approximately 30 people you need to vaccinate in this age group to prevent one case of shingles over a three-year period.
HOW TO READ NNT
If the NNT is 30, it means that for every 30 people vaccinated, 29 received no direct benefit from the vaccine over the trial period (because they wouldn’t have developed shingles anyway), and 1 was protected from a shingles case. The other 29 still took on whatever risks or side effects the vaccine entailed. NNT doesn’t tell you which person you’ll be; it tells you the odds.
How NNT varies by age
One of the genuinely useful features of Shingrix is that its relative efficacy doesn’t fall off with age the way the older vaccine did. The percentage reduction in risk is roughly similar whether you’re 55 or 80.
But, and this is important, the absolute benefit varies by age because older people face a higher baseline risk of shingles. More cases to prevent means more absolute benefit per vaccination

Notice: the relative efficacy is almost identical across all ages. But the absolute numbers diverge because the baseline disease rate varies. A 97% reduction in a low-risk group gives you less absolute benefit than a 91% reduction in a high-risk group.
This is not a trick or a critique of the vaccine; it’s just how mathematics works. But it does mean that when you read “97% effective,” you should immediately ask: 97% of what? Of whom? Over how long?
THE SIDE EFFECTS
The side effects from the vaccine are real, and the trials likely undercounted them
Shingrix has a strong adjuvant, a chemical booster that makes your immune system respond more intensely. This is how it achieves such high efficacy even in older adults. The tradeoff is a higher rate of side effects than most vaccines.
In the trials, 78 to 84 percent of vaccinated people reported at least one reaction within 7 days, compared with about 30 percent in the placebo group. Most were mild: sore arm, tiredness, headache. But a meaningful proportion were severe enough to disrupt daily life. This is measured as “grade 3” reactions — fever over 39°C, muscle pain that prevents normal activity, and injection-site pain at rest.
The “number needed to harm” (NNH) for a grade 3 reaction, meaning one serious enough to stop you doing normal things for a day or two, was:
• Ages 50–69: approximately 1 in 11 vaccinated people
• Ages 70+: approximately 1 in 25 vaccinated people
Older adults have fewer severe reactions, not because the vaccine is gentler, but because their immune systems respond less intensely to stimulation. This is, slightly counterintuitively, the same reason the relative efficacy is a few percentage points lower in the oldest group.
Here’s the comparison that actually matters: across the three-year trial window, for every case of shingles prevented in the 60–69 age group, approximately 2.7 people experienced a grade 3 reaction. Those reactions lasted one to two days. Shingles and its complications can last far longer.
However, there is an important asterisk on all the side effect data: the trials were not fully blinded.
THE BLINDING PROBLEM
In a fully “double-blind” trial, neither the participant nor anyone treating them knows whether they received the real drug or the placebo. Shingrix couldn’t achieve this because the vaccine, when mixed, looks visibly different from clear saline. The nurse preparing the syringe knew what they were drawing up.
More importantly, the vaccine causes such distinctive reactions, a cluster of fever, fatigue, and muscle aches within 24 hours, that many participants who received it almost certainly figured out they were in the vaccine group. Research consistently shows that people who know (or strongly suspect) they got the active treatment tend to report more symptoms on diary cards. This means the side-effect numbers from the trial are probably somewhat higher than they would be in a perfectly blinded study. No one knows by how much.
THE EVIDENCE TRAIL
Every single trial was paid for by the company selling the vaccine
This is worth saying plainly, without drama: all of the clinical evidence for Shingrix, every pivotal trial, every extension study, every long-term follow-up analysis, was funded by GlaxoSmithKline, the vaccine’s manufacturer. Several named authors on the key publications were GSK employees at the time. Medical writing for the main publications was paid for by GSK. The decision about when to submit results for publication was made by GSK.
None of this is unusual. This is standard practice in the pharmaceutical industry, and it’s the only realistic way to fund trials of this size. But it does mean you’re relying on a company with a direct financial interest in a positive result for every piece of evidence about the product’s efficacy and safety. No independent, publicly-funded pivotal trial of Shingrix exists.
Industry funding doesn’t automatically mean the results are wrong. The trials used randomization, independent adjudication panels, and safety monitoring committees. Post-marketing studies conducted without GSK involvement have found broadly similar effectiveness figures. But those corroborating studies have their own limitations, and “broadly similar” still means lower effectiveness than the headline trial numbers. Real-world effectiveness tends to be 10 to 15 percentage points lower than controlled-trial efficacy because real patients are older, sicker, and on more medications than the trial cohort.
The long-term data has a significant flaw
GSK extended the trials with a follow-up study called ZOE-LTFU, which tracked participants for up to 11 years post-vaccination. The results looked good: roughly 88% relative efficacy maintained through year 11.
The problem: by the time the extension started, the original placebo group had already been offered the vaccine (because the main trial was over). There were no unvaccinated controls left. So the researchers had to calculate efficacy by comparing vaccinated participants against a projected placebo group based on earlier data, essentially asking: how many cases would we have expected if these people had never been vaccinated, using the old placebo rates?
This approach has real limitations. It assumes shingles rates didn’t change over time. It doesn’t account for the COVID-19 pandemic (which affected immune system dynamics and social mixing). It assumes the people who stayed in the study for 11 years are representative of everyone who started. None of these assumptions can be verified. GSK itself acknowledged in the publication that this methodology “could overestimate efficacy.”
IF YOU DO GET SHINGLES
The treatments for shingles work, if you get them quickly
If you do develop shingles, three antiviral medications: valacyclovir, famciclovir, and acyclovir, can reduce its severity and duration if you start them within 72 hours of the rash appearing. That window matters a great deal. After 72 hours, the benefit drops off significantly.
These antivirals reduce the severity of the shingles episode but don’t reliably prevent PHN. Once that nerve pain develops, treatment becomes much harder.
IF YOU DEVELOP PHN
The first-line treatments are gabapentin and pregabalin (anticonvulsants used as nerve pain drugs), tricyclic antidepressants like nortriptyline, and lidocaine patches applied to the skin. These help some people meaningfully. But approximately 40 to 50 percent of PHN patients don’t get adequate relief from any of them. Opioids are sometimes used but carry dependence risks that are especially significant in older adults. There is no cure.
THE BOTTOM LINE
So, should you get it?
This content is for paid subscribers only: