Congress Builds a Safety Net (Part 2)
The Creation of OPPR, the Constitutional Limits of Federal Health Authority, and What the Office Actually Accomplished
The state is that great fiction by which everyone endeavors to live at the expense of everyone else.
Frédéric Bastiat, “The State,” 1848
Summary
Congress’s response to the COVID-19 coordination catastrophe was the PREVENT Pandemics Act, signed into law in December 2022 as part of the omnibus appropriations bill. Section 2104 mandated the creation of the Office of Pandemic Preparedness and Response Policy within the Executive Office of the President, with a presidentially appointed director, biennial reporting obligations to Congress, and a five-year preparedness outlook. The Act reflected a genuine conservative public health tradition: the federal government has a legitimate role in maintaining readiness for catastrophic biological threats, just as it has a legitimate role in maintaining military readiness. Preparedness is not the same as intervention.
This installment places OPPR’s creation within the constitutional architecture that Dobbs v. Jackson and NFIB v. OSHA clarified: the federal government has no general police power over health; direct control of medical practice in the states is beyond its authority; and the proper role of federal pandemic institutions is to support and coordinate, not to assume operational control. It examines the NSC predecessor function performed by Raj Panjabi, whose simultaneous role in WHO Pandemic Accord negotiations raises sovereignty concerns addressed in full. And it documents what OPPR actually did during its two years of operation: the H5N1 interagency coordination that FDA Commissioner Califf credited in Senate testimony, the Moderna vaccine contract, the dose-fractionation decision during the 2022 mpox response, the Bio-5 Biopharmaceutical Alliance, and the Biological Incident Response Playbook.
The operational record matters because it is the evidence base for the argument made in Part Three: that the Trump administration’s attrition of a functioning statutory institution is not conservative governance. Before that argument can be evaluated, the record of what OPPR built must be established. This installment provides that record.
PART TWO — SECTION A
Congress Creates OPPR: The Political Structure of a Pandemic Bureaucracy
The Legislative Response to COVID-19’s Coordination Failures
The COVID-19 pandemic exposed a specific and correctable structural failure in the federal government’s response architecture: there was no single entity within the Executive Office of the President whose specific statutory mandate was to coordinate the interagency response to a biological threat across all relevant federal departments simultaneously. The CDC had surveillance authority. HHS had emergency declaration authority. FEMA had logistics authority. The NSC had national security coordination authority. BARDA had medical countermeasure development authority. No single office had the mandate and the position to convene all of them around a unified operational picture on a standing basis.
The result, which COVID-19 demonstrated in real time, was that interagency coordination happened through ad hoc structures, bilateral negotiations between agencies, and emergency mechanisms that had to be assembled from scratch as the crisis evolved. Critical decisions about vaccine distribution, testing capacity, and countermeasure procurement were made under conditions of incomplete information because no entity had the statutory role of aggregating what each agency knew into a coherent operational picture. The PREVENT Pandemics Act was Congress’s bipartisan diagnosis of this specific failure and its prescription for correcting it.
The Congressional Architecture: What the Statute Actually Required
The PREVENT Pandemics Act (Pub. L. 117-328, Section 2104) was signed into law on December 29, 2022, as part of the Consolidated Appropriations Act. The relevant provisions mandated the establishment of the Office of Pandemic Preparedness and Response Policy within the Executive Office of the President. The office’s director was to be presidentially appointed, providing it with institutional standing to convene interagency processes that a career official or a detail from an existing agency would not have had. The statutory mandate included: serving as the principal adviser to the President on pandemic preparedness; coordinating federal activities related to pandemic preparedness and response policy; engaging with state, local, tribal, and territorial governments, as well as international partners; and overseeing implementation of the National Biodefense Strategy.
The Act required the OPPR director to submit a biennial preparedness review to Congress and a five-year preparedness outlook. These reporting requirements were not bureaucratic formalities. They were the mechanism by which Congress attempted to ensure that pandemic preparedness remained a visible, accountable government function rather than a periodic emergency reaction. A White House office that was required to report its findings to Congress every two years had an institutional incentive to have something to report, and Congress, which received those reports, had the information basis for the appropriations and oversight decisions mandated by the statute.
Bipartisan Origins and Conservative Ambivalence
The PREVENT Pandemics Act passed with significant Republican support. Senator Richard Burr of North Carolina, the ranking Republican on the Senate Health Committee, was among its key architects. The legislation reflected a genuine conservative public health tradition: the belief that government has a legitimate role in preparing for catastrophic biological threats, in the same way that it has a legitimate role in maintaining military readiness or flood control infrastructure. Preparedness is not the same as intervention. Building the capacity to respond to a pandemic is not the same as running the pandemic response in ways that override individual liberty
.Conservative ambivalence about OPPR stems from a different source: the concern that creating a permanent White House office dedicated to pandemic preparedness will inevitably tend toward mission creep, expanding government authority, and the kind of permanent bureaucratic class that develops institutional interests in the perpetuation and expansion of its own mandate. These concerns are not irrational. The Biden administration’s management of the COVID-19 Response Team demonstrated precisely how pandemic-related authority can be weaponized for political purposes. The question is whether the answer to that risk is better oversight and clearer statutory limits, or the abandonment of the institutional capacity entirely.
Dobbs v. Jackson and the Constitutional Architecture of Federal Medical Authority
The conservative case for limiting federal pandemic authority is not merely a political preference. It is, as the Supreme Court has made increasingly clear, a constitutional argument with growing doctrinal weight. No decision has clarified the constitutional architecture of federal medical authority more starkly in recent years than Dobbs v. Jackson Women’s Health Organization, 597 U.S. 215 (2022). Dobbs is understood primarily as an abortion case. Its implications for the structural question of who governs medicine in America extend considerably further.
The Dobbs majority opinion, written by Justice Samuel Alito and joined by four other justices, located the abortion question within a broader principle that has direct application to pandemic governance. The Court held that the Constitution does not confer a right to abortion and returned the regulation of abortion to the states. In doing so, it restated a foundational constitutional proposition that had been obscured by decades of federal expansion into health policy: that ‘direct control of medical practice in the states is beyond the power of the federal government,’ and that the federal government possesses no general police power over health, education, and welfare. That power belongs, by constitutional design, to the states.
Dobbs restated a foundational constitutional proposition: ‘direct control of medical practice in the states is beyond the power of the federal government.’ The federal government has no general police power over health.
This is not a novel constitutional claim. It is a restatement of the Tenth Amendment’s structural logic, applied to a specific and consequential domain. The states hold plenary police power over public health under the constitutional design. The Tenth Amendment reserves to the states all powers not delegated to the federal government by the Constitution. The federal government has no enumerated power over health and medicine; it acts in the health domain through the Commerce Clause, the Spending Clause, and specific statutory delegations, each of which has limits. Jacobson v. Massachusetts, the foundational public health federalism case from 1905, upheld a state vaccine mandate, not a federal one, precisely because the Court recognized that this kind of regulatory authority belonged to the states and their police power, not to the national government.
The COVID-19 pandemic put this constitutional architecture under severe stress. The Biden administration’s approach to pandemic management was built on an expansive theory of federal authority: that a sufficiently severe public health emergency could sustain federal vaccine mandates on private employers via OSHA, federal eviction moratoriums via CDC, and a sustained White House operation to shape the national information environment. The Supreme Court pushed back at each turn. In National Federation of Independent Business v. OSHA (2022), decided the same term as Dobbs, the Court blocked the Biden administration’s large-employer vaccine-or-test mandate, holding that OSHA had no authority to impose a sweeping health regulation that went far beyond workplace-specific hazard control. The Court explicitly invoked the major questions doctrine: when an agency claims authority of vast economic and political significance, it must point to clear congressional authorization. Pandemic management is not a clear OSHA authorization. Pandemic management is, constitutionally, primarily a state function.
The implication for OPPR and for the broader apparatus of federal pandemic preparedness is uncomfortable but important. The conservative case for limiting OPPR’s mandate is not simply a preference for smaller government. It is a constitutionally grounded argument that the federal government’s proper role in health emergencies is narrow, specific, and defined by statutory authorization rather than executive ambition. The federal government can maintain vaccine stockpiles, fund research, and support logistics that only national resources can provide. What it cannot constitutionally do, as Dobbs, NFIB v. OSHA, and the vaccine mandate cases collectively make clearer, is assume general supervisory authority over the medical decisions of states, institutions, and individuals.
The Dobbs framework also reinforces a point about democratic legitimacy that runs throughout the conservative critique of the COVID-19 response. The Court’s holding was explicitly that profound questions about medical practice and bodily autonomy should be resolved by the people’s elected representatives in the states, not by federal courts or federal agencies. That principle applies with equal force to pandemic governance. The decisions made during COVID-19 about mask mandates, school closures, business restrictions, and vaccine requirements were of exactly the kind of local political significance that, under the constitutional design, should be made by governors and state legislators accountable to their citizens, not by White House officials accountable to no one outside the executive branch.
The federalism argument also has a practical dimension that the COVID-19 experience validates. The patchwork of state responses to COVID-19, widely criticized by centralizing commentators as inconsistent, also served as a set of natural policy experiments. States that took different approaches to school reopening, business restrictions, and masking generated data that, over time, permitted more evidence-based assessment of which interventions actually affected outcomes and at what cost. A fully federalized pandemic response, in which Washington determined the single national approach, would have foreclosed that comparative learning. The states’ traditional role as laboratories of democracy is not merely a federalism abstraction. In public health, as in other policy domains, it is a genuine epistemic resource.
None of this is an argument against federal pandemic-preparedness investment of the kind that OPPR was designed to coordinate. Federal funding for vaccine development, stockpile management, and international surveillance is constitutionally grounded in enumerated powers and practically justified by the national-security character of pandemic preparedness. The Dobbs framework does not prohibit federal spending on public health. What it does is clarify where the constitutional boundary lies: between federal preparedness support and federal operational control of medical practice. OPPR, properly understood, was designed to sit on the permissible side of that line. The Biden administration’s COVID-19 Response Team operated well past it. The conservative task is to hold that distinction rigorously and resist the institutional tendency to let preparedness authority expand into operational control during the next emergency, when the political pressure to do so will be overwhelming.
Raj Panjabi: The NSC Predecessor and the WHO Connection
The official who most directly shaped Biden’s pandemic preparedness framework before OPPR existed was Raj Panjabi, who served as White House Senior Director for Global Health Security and Biodefense at the National Security Council from 2021 to 2023. Panjabi’s biography is worth knowing: born in Liberia in 1981 to Indian immigrant parents, he fled civil war at age nine and eventually earned his MD from the University of North Carolina, an MPH in epidemiology from Johns Hopkins, and completed clinical training at Massachusetts General Hospital and Harvard Medical School. In 2007 he co-founded Last Mile Health with $6,000 in wedding gifts, eventually training approximately 16,000 community health workers serving 19 million people across Liberia, Ethiopia, Malawi, and Sierra Leone. He received a 2017 TED Prize and was knighted by Liberian President Ellen Johnson Sirleaf.
At the NSC, Panjabi coordinated more than $12 billion in annual federal biodefense investment across 16 agencies, led implementation of the 2022 National Biodefense Strategy, and represented the United States in negotiations over both the WHO International Health Regulations and the WHO Pandemic Accord. That last role requires scrutiny. The WHO Pandemic Accord, finalized in 2024 after years of contentious negotiation, contains provisions that critics across the political spectrum have identified as problematic: pathogen access and benefit-sharing arrangements that could compel nations to share biological samples and intellectual property with the WHO secretariat; a proposed surveillance framework granting WHO authorities to declare pandemics that trigger member-state obligations; and equity provisions requiring wealthy-nation pharmaceutical companies to donate a portion of pandemic vaccine production. Panjabi also served as technical adviser to former President Sirleaf as co-chair of the WHO Independent Panel for Pandemic Preparedness and Response, whose 2021 report ‘COVID-19: Make it the Last Pandemic’ recommended establishing a new Global Health Threats Council at head-of-government level with mandatory financing authority, a proposal that conservatives argued would transfer meaningful national sovereignty over health emergencies to an unaccountable international body. These are not marginal concerns. The official who designed Biden’s domestic pandemic preparedness architecture was simultaneously the administration’s primary representative in negotiations over the international governance framework that conservatives have consistently identified as the most significant long-term threat to U.S. health sovereignty.
The NSC location of Panjabi’s role had an institutional significance that Congress deliberately addressed through OPPR. NSC records are shielded from congressional oversight and public FOIA access by the Presidential Records Act. A pandemic preparedness function housed there operates largely outside public view. OPPR, placed within the broader Executive Office of the President, was designed to provide what the NSC cannot: statutory accountability, congressional reporting requirements, and public transparency. When the Trump administration declined to staff OPPR and effectively returned preparedness functions to the NSC, it replicated precisely the structural opacity Congress had tried to correct. After leaving government in 2023, Panjabi joined Flagship Pioneering as Senior Partner, launched the AI health venture Etiome, and co-signed bipartisan CSIS briefs with inaugural OPPR director Paul Friedrichs urging the Trump administration to reverse its attrition of the biodefense enterprise. He serves on the boards of the WHO Foundation and the Bipartisan Commission on Biodefense.
OPPR’s Launch, Staffing, and the Budget Problem
OPPR formally launched in July 2023. Major General (ret.) Paul Friedrichs was named inaugural director. A physician and retired Air Force officer who had served as Joint Staff Surgeon and medical adviser to the Department of Defense’s COVID-19 Task Force, Friedrichs was a credible, non-partisan choice who had spent a career in military medicine and interagency coordination. Nikki Romanik served as Deputy Director and Chief of Staff. The office eventually grew to more than twenty experts drawn from medicine, science, policy, and defense.
Almost immediately, OPPR confronted a structural problem that its congressional architects had not fully resolved: the office was mandated without a dedicated appropriation. Congress required OPPR to exist but did not designate specific funding for it. The government’s reliance on a series of continuing resolutions rather than regular appropriations meant that the $6.8 million Friedrichs estimated was needed to fully perform the office’s statutory functions was never actually appropriated. OPPR operated, in Friedrichs’s later description, on a shoestring.
This underfunding is itself a congressional accountability failure worth noting. Congress created a statutory mandate but failed to ensure that the appropriations process followed through. An office that cannot independently hire, contract, or build institutional momentum is perpetually dependent on the White House’s discretionary support, which means its effectiveness tracks the current administration’s level of interest in pandemic preparedness rather than the statutory mandate Congress established.
PART TWO — SECTION B
OPPR in Operation: Monkeypox, H5N1, and the Infrastructure of Preparedness
Demetre Daskalakis and the 2022 Mpox Response: The Problem OPPR Was Designed to Solve
The 2022 mpox outbreak arrived before OPPR existed, and the federal government’s handling of it documented in real time the coordination failures Congress sought to correct through the PREVENT Pandemics Act. President Biden appointed FEMA Regional Administrator Robert Fenton as White House National Monkeypox Response Coordinator and Dr. Demetre Daskalakis, then CDC Director of HIV/AIDS Prevention, as deputy coordinator on August 2, 2022. The appointments came nearly three months after the first confirmed U.S. case. By that date more than 5,800 cases had accumulated and no federal public health emergency had yet been declared; the declaration followed two days later. A GAO audit released in 2024 concluded that HHS’s response had been uncoordinated and lacked interagency cohesion, precisely the structural deficit OPPR was designed to remedy
.The most consequential and contested policy decision of the Daskalakis-led response was the August 9 FDA Emergency Use Authorization for intradermal dose-fractionation of the JYNNEOS vaccine. JYNNEOS had been approved in 2019 for smallpox prevention in adults at high risk, with a separate monkeypox indication added as an extension of that approval. It had been stockpiled, in minimal quantities, for smallpox bioterrorism contingencies, not for deployment against a sexually transmitted outbreak in a population of potentially one to two million high-risk individuals. The United States entered August 2022 with roughly 440,000 doses on hand against a demand that vastly exceeded supply. The administration’s solution was dose fractionation: a one-fifth subcutaneous dose administered intradermally, between the skin layers rather than beneath them, on the basis of a single 2015 immunogenicity study showing comparable antibody response at the fractional dose. The EUA quintupled effective supply overnight. FDA Commissioner Robert Califf was candid about the evidentiary basis: there was no traditional clinical outcomes assessment for the vaccine because prior monkeypox outbreaks had not been large enough to support one. The administration was deploying a smallpox vaccine at a novel dose and route, in a novel indication, justified by immunological proxy data rather than efficacy trials. A May 2023 breakthrough cluster in Chicago among fully vaccinated individuals subsequently raised durability questions that CDC research left unresolved. Daskalakis appeared at the White House podium to defend the strategy and served as the administration’s primary interface with LGBTQ+ community organizations throughout the response. When OPPR launched in July 2023, he had returned to CDC as Director of the National Center for Immunization and Respiratory Diseases, where the relationships he had formed with OPPR’s Nikki Romanik and Paul Friedrichs during the mpox deployment would later facilitate coordination during the H5N1 response.
The Monkeypox Response: OPPR and the Clade I Outbreak
OPPR’s own mpox test came with the Clade I outbreak, which required coordinated domestic and international response across multiple federal agencies with overlapping but distinct authorities. The CDC held primary authority for domestic disease surveillance and public health guidance. The FDA regulated vaccines and treatments. USAID and the State Department managed international dimensions. HHS’s Office of the Assistant Secretary for Preparedness and Response (ASPR) coordinated medical countermeasures. Without a central convening authority, these agencies would coordinate bilaterally, slowly, and incompletely.
OPPR served as that central convening authority. The office established an operational structure that brought agencies together under a unified situational picture, identified gaps in the federal response, and helped prevent the kind of inter-agency disputes over authority and resources that had characterized COVID-19’s early months. OPPR also coordinated with the U.S. Agency for International Development and international partners, ensuring that domestic and international response efforts were not working at cross-purposes.
The mpox response also demonstrated OPPR’s capacity to work with the Strategic National Stockpile. ASPR manages the stockpile, but OPPR could identify the countermeasures needed, assess gaps between stockpile holdings and response requirements, and work across agencies to address those gaps before they became critical. This pre-positioned coordination work, done before an emergency requires maximum response speed, is precisely the kind of institutional function that has no visible constituency but proves its value when the emergency arrives.
H5N1: The Most Consequential Test
OPPR’s most sustained and consequential operational challenge involved the H5N1 avian influenza outbreak in American dairy cattle, which began in the spring of 2024 and remained an active concern at the time of the Trump administration’s transition. The H5N1 case illustrates both OPPR’s genuine operational value and the difficult policy questions that arise when a public health threat intersects with agricultural economics and individual liberty concerns.
The outbreak presented an unusual interagency challenge. H5N1 is a pathogen that intersects animal health, human health, food safety, and agricultural economics. The United States Department of Agriculture, through its Animal and Plant Health Inspection Service, had primary authority over infected livestock and the federally mandated testing and movement controls for dairy cattle. The Food and Drug Administration was responsible for the safety of the commercial milk supply and had authority over pasteurization standards. The CDC tracked human cases among dairy workers and managed the public health surveillance infrastructure. The NIH drove the underlying research on viral characteristics, transmission dynamics, and vaccine development. The National Institutes of Health and BARDA, the Biomedical Advanced Research and Development Authority, managed the medical countermeasure pipeline.
No single agency had the authority or the visibility to coordinate all of these functions simultaneously. OPPR did. Beginning in March 2024, Director Friedrichs established daily interagency calls that brought together senior representatives from USDA, FDA, CDC, NIH, ASPR, HHS, and the VA. The calls moved to three times per week as the situation stabilized, and eventually to weekly. FDA Commissioner Robert Califf credited OPPR in testimony before the Senate Appropriations Committee for coordinating the response “at the highest level,” a recognition that the office was performing a function that no individual agency could replicate.
Medical Countermeasures: The Moderna Contract and Vaccine Development
One of OPPR’s most significant specific accomplishments in the H5N1 response was its management of the medical countermeasure pipeline. Vaccine development for novel or mutating pathogens is unavoidably time-intensive. A pathogen can evolve from manageable to catastrophic in a matter of months. The only way to ensure a ready-to-deploy vaccine exists when it is needed is to fund the development work before the emergency arrives.
OPPR coordinated two BARDA awards to Moderna totaling $766 million: an initial $176 million in July 2024 and a subsequent $590 million in January 2025, to develop mRNA-1018, an mRNA-based vaccine candidate against H5N1. The January award was specifically intended to fund a late-stage clinical trial that could have established the vaccine’s efficacy against pandemic influenza strains. OPPR also worked with BARDA to maintain a diverse portfolio of H5N1 vaccine candidates across multiple technology platforms, recognizing that any single vaccine program might encounter manufacturing, safety, or efficacy obstacles. The mRNA platform’s speed advantage, demonstrated during COVID-19, made it a particular priority for next-generation H5N1 preparedness.
The Trump administration cancelled the full $766 million on May 28, 2025. The cancellation was announced the same day Moderna released positive interim Phase 1/2 data showing mRNA-1018 was well-tolerated and produced a rapid immune response in approximately 300 healthy adults, with 98 percent achieving antibody titers within three weeks of the second dose. HHS Communications Director Andrew Nixon stated that continued investment was not scientifically or ethically justifiable, citing concerns about the mRNA platform’s safety profile and mounting evidence of adverse events associated with COVID-19 mRNA vaccines. Nixon did not cite peer-reviewed literature in support of that characterization. Existing egg-based and cell-based H5N1 vaccine candidates remain in the federal pipeline.
Beyond vaccines, OPPR coordinated with the VA and FEMA to maintain therapeutics in the Strategic National Stockpile for deployment if H5N1 transmission among humans increased. This included antivirals active against influenza strains, personal protective equipment for healthcare workers in dairy and poultry operations, and diagnostic capacity sufficient for rapid surge testing if human cases began to multiply. OPPR also worked with industry partners to pre-position manufacturing agreements to rapidly scale up countermeasure production if needed.
The Government-Wide Countermeasure Mapping Project
One of OPPR’s most consequential long-term contributions was less visible than its crisis coordination work: a comprehensive, government-wide inventory of U.S. investments in medical countermeasures across all biological threat categories. Before this exercise, no single entity within the federal government had a complete picture of what medical countermeasures existed in the stockpile, what was in development across federal research programs, what gaps remained, and where the most urgent R&D priorities lay.
The mapping project identified critical vulnerabilities, particularly in the pipeline for next-generation countermeasures against pathogens with pandemic potential. It revealed areas where federal investment was duplicative and areas where it was absent. The exercise helped OPPR redirect federal R&D priorities toward emerging threats, including H5N1, and established a framework for continuing this kind of systematic assessment. The resulting picture of the nation’s countermeasure portfolio was, in a meaningful sense, a national security asset: the United States could not effectively prepare for what it could not clearly inventory.
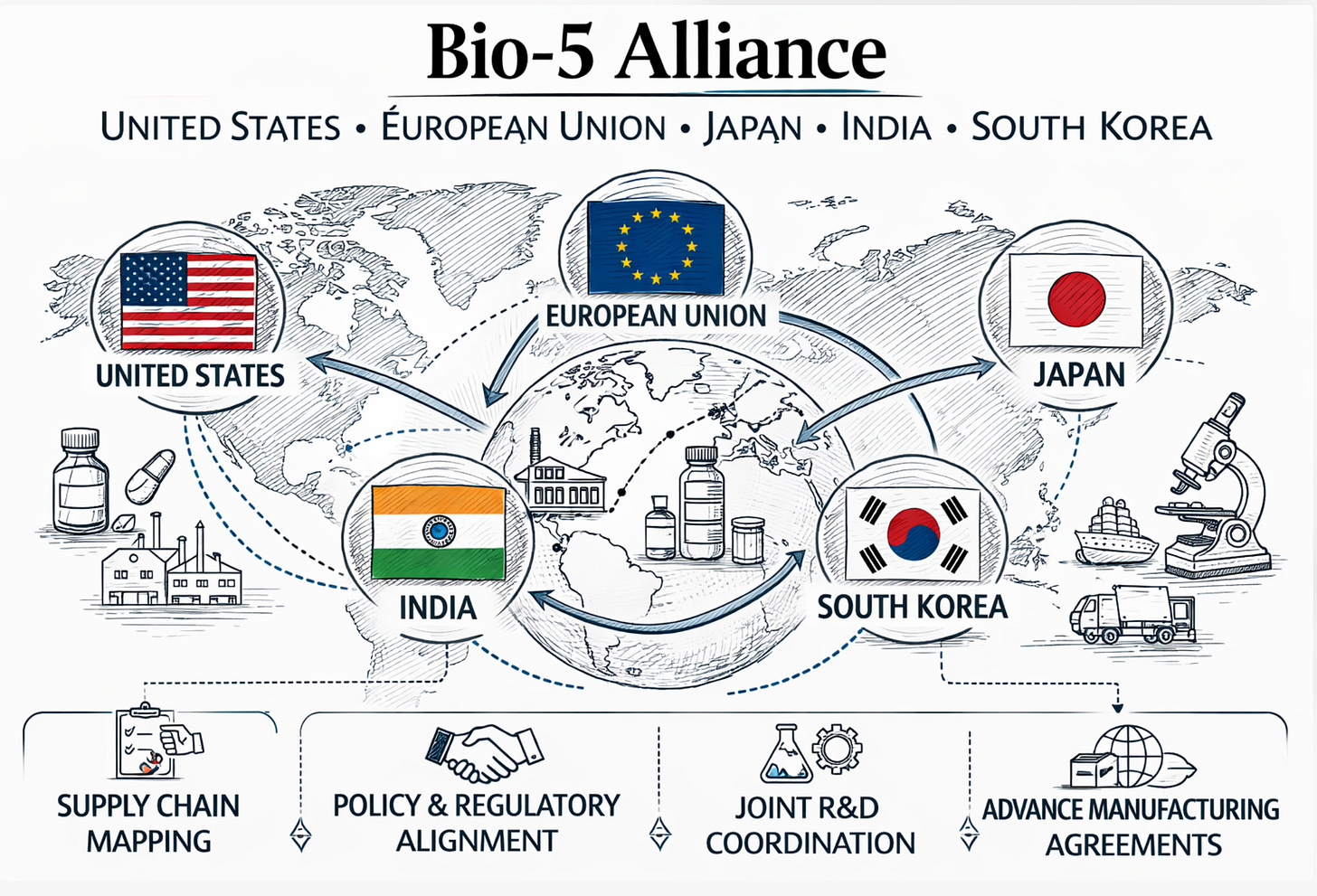
The Bio-5 Alliance: International Supply Chain Security
OPPR’s international work extended beyond coordination with foreign governments on disease surveillance. In June 2024, OPPR worked with the National Security Council to formally announce the Bio-5 Biopharmaceutical Alliance, a partnership among the United States, the European Union, Japan, India, and the Republic of Korea. The alliance’s inaugural meeting took place during the BIO International Convention in San Diego, the world’s largest biotechnology gathering, with government officials and private-sector representatives from all five parties in attendance. The coalition’s genesis traced to a bilateral dialogue between the United States and South Korea on core emerging technologies in December 2023; that conversation expanded to include Japan, India, and the EU as the scope of the supply chain problem became clear. The five members collectively represent the world’s major democratic pharmaceutical manufacturing economies, and notably India, the world’s largest generic drug producer and a primary source of active pharmaceutical ingredients for the U.S. market.
COVID-19 demonstrated that a global pandemic does not simply create demand for medical countermeasures; it simultaneously disrupts the supply chains needed to manufacture and distribute them. Active pharmaceutical ingredients for many essential drugs are manufactured in a small number of countries, often concentrated in the People’s Republic of China. The inaugural Bio-5 meeting explicitly acknowledged this: participants recognized that essential raw materials and ingredients were concentrated in too few countries and agreed that reducing that concentration was a shared national security priority. The BIOSECURE Act, which sought to restrict Chinese biotech firms from integrating with the U.S. medical supply chain, reflected the same diagnosis on the legislative track.
The Bio-5 Alliance’s political structure operates on a track 1.5 format, meaning that government officials and private industry representatives participate jointly, under the auspices of the NSC and OPPR on the U.S. side, with equivalent governmental counterparts from each member economy. This structure was chosen deliberately: supply chain resilience cannot be achieved by government mandates alone; it requires advance commitments from the pharmaceutical manufacturers, contract development and manufacturing organizations, and raw material suppliers who would actually execute diversified production arrangements. The format gives governments and industry a shared forum to align policy incentives with commercial investment decisions before a crisis makes that alignment urgent.
The alliance’s mission tasks fell into four categories. First, supply chain mapping: member countries agreed to develop a comprehensive pharmaceutical supply chain map identifying critical nodes, concentration risks, and potential points of failure, beginning with active pharmaceutical ingredients sourced primarily from the PRC. Second, policy and regulatory harmonization: participants agreed to coordinate bio policies, regulations, and R&D support measures to reduce bureaucratic friction that impedes the surge capacity for cross-border manufacturing under emergency conditions. Third, joint R&D coordination: members committed to pooling resources and expertise to accelerate innovation, with particular attention to the gap between laboratory development and commercial-scale manufacturing that had hampered countermeasure deployment during COVID-19. Fourth, advance manufacturing agreements: the alliance sought to develop standing commitments for mutual production assistance that could be activated during declared emergencies, so that allied manufacturing capacity could be directed toward the most affected partner rather than commandeered by each government for purely domestic use. Whether the alliance survives the abandonment of OPPR, and whether the bilateral and multilateral relationships it established remain functional under the Trump administration’s different approach to allied health security coordination, is a question the available record does not yet answer.
The Biological Incident Response Playbook
Shortly before the Biden administration ended, OPPR completed the Playbook for Biological Incident Response, a formal interagency document developed in collaboration with the NSC’s Biosecurity and Pandemic Response directorate. The playbook operationalized the lessons of COVID-19 and the H5N1 response into a standing set of procedures for rapidly coordinating the U.S. government’s response to biological threats.
The playbook specified decision authorities, communication protocols, agency roles and responsibilities, and escalation procedures across a range of biological threat scenarios. Critically, it established a common operational picture framework that would allow agencies to share situational awareness rapidly without the interagency disputes over data ownership and interpretation that had complicated the COVID-19 response. The Clade I Mpox response had served as a real-world validation of the playbook’s underlying framework; adjustments made during that response were incorporated into the final document.
The playbook represents a specific kind of institutional asset that takes years to develop and can be destroyed in weeks. The interagency relationships, the shared vocabulary, the established protocols, the tested escalation procedures: these are not things that can be recreated rapidly under emergency conditions by officials who have never worked together through a functional preparedness structure. The value of the playbook is not the document itself. It is the institutional knowledge embedded in the people who developed it and who understand how to use it. Those people have left the government.
Congressional Reporting and the Preparedness Review
Among OPPR’s statutory obligations were the biennial Preparedness Review and five-year Preparedness Outlook reports to Congress. OPPR prepared these reports under the Biden administration, delivering assessments that identified specific vulnerabilities in biosurveillance, supply chain resilience, medical countermeasure development, and international coordination. The reports reflected OPPR’s comprehensive countermeasure mapping work and provided Congress with a systematic basis for evaluating federal preparedness investments in appropriations proceedings.
The reports are no longer being prepared. The statutory obligation exists. The office that would fulfill it does not exist in practice. Congress is therefore being denied information that an Act of Congress requires to be provided. This is not a minor compliance gap. The reporting requirement was the primary mechanism by which Congress could hold the executive branch accountable for the quality of its pandemic preparedness without waiting for the next emergency to reveal what the government had failed to do.
A Congress that does not receive its statutorily required preparedness reports and does not respond to that failure with the tools available to it, including appropriations conditions, hearing subpoenas, and public accountability proceedings, is a Congress that has quietly acquiesced in the attrition of a function it created and mandated. The blame for the current state of OPPR is not the executive branch’s alone.
Sources and Notes
This series draws on: court filings and discovery documents in Missouri v. Biden / Murthy v. Missouri (W.D. La. 2022, 5th Cir. 2023, S. Ct. 2024); the House Judiciary Committee’s report “The Censorship-Industrial Complex” (2024); Missouri Attorney General’s litigation documents; reporting by the Wall Street Journal, CNN, STAT News, the Washington Times, and National Review; testimony before the House Select Subcommittee on the Coronavirus Pandemic (March 2024) and the House Weaponization of the Federal Government Subcommittee (May 2024); official White House fact sheets and archived Biden White House pages; the Supreme Court’s opinion in Murthy v. Missouri, 603 U.S. 43 (2024); the AEI op-ed co-authored by former OPPR staff (August 2025); and Think Global Health analysis of OPPR’s demise (2025). Direct quotations from emails are drawn from court documents and congressional records.







Regarding conservatives attitude...I groaned when Bush created DHS...we needed another agency like the proverbial pimple on the tush. We already had too many as the Waco incident pretty well demonstrateded. What was needed was that agencies like ATF and DEA had never been formed and short of that redundant functions consolidated into fewer agencies rather than creating a new one. And I strongly suspect the same could be said about the various health related federal agencies. Perhaps consolidation and reduction would have been a better approach. The fed is too damn big and does not need to keep getting bigger
The Good, The Bad And The Ugly, / all in one package. Trying to keep these agencies in a coordinated fashion is like trying to herd cats...nearly impossible. There are some extremely intelligent minds at work here, which certainly doesn't make the integrity of the process and product any more accountable, as we have seen. Somehow the influence of the seven deadly sins creeps in. Just how deep the creep is to be seen.