Immune Imprinting, Immunosenescence, and the Limits of Universal Seasonal Influenza Vaccination
A Critical Review of Two Decades of Vaccine Effectiveness Surveillance Data in the United States
Immune Imprinting, Immunosenescence, and the Limits of Universal Seasonal Influenza Vaccination: A Critical Review of Two Decades of Vaccine Effectiveness Surveillance Data in the United States
What Is Already Known About This Topic?
Seasonal influenza vaccination is universally recommended for all persons aged >=6 months in the United States. Standard-dose inactivated vaccines have demonstrated variable overall vaccine effectiveness (VE) ranging from 10% to 60% since systematic monitoring began in 2004.
What Is Added by This Report?
This report synthesizes two decades of VE surveillance data with mechanistic evidence for immune imprinting and immunosenescence, demonstrating that host immune factors -- particularly childhood influenza-subtype exposure and age-related immune decline -- substantially modify VE in ways not accounted for by current universal vaccination policy.
What Are the Implications for Public Health Practice?
Universal seasonal influenza vaccination policies may benefit from reassessment in light of evidence that repeat vaccination attenuates VE, particularly for influenza A(H3N2), and that standard-dose vaccines provide suboptimal protection in older adults. Risk-stratified or platform-stratified approaches warrant evaluation.
Abstract
Universal seasonal influenza vaccination has been recommended for all persons aged >=6 months in the United States since 2010. However, two decades of vaccine effectiveness (VE) surveillance demonstrate that overall adjusted VE has ranged from 10% to 60%, with a mean near 40%, reflecting persistent and largely unexplained variation. Emerging evidence implicates two host immunological phenomena -- immune imprinting (the lifelong bias in immune response imposed by the first influenza subtype encountered in childhood) and immunosenescence (the age-related deterioration of innate and adaptive immune function) -- as major, underappreciated determinants of VE that are not addressed by current universal vaccination policy. This report synthesizes two decades of CDC Influenza Vaccine Effectiveness Network data alongside mechanistic and epidemiological evidence for both phenomena. The data suggest that (1) repeat annual vaccination attenuates VE, particularly against influenza A(H3N2), through back-boosting of outdated immune memory; (2) childhood subtype imprinting shapes adult susceptibility in ways that are independent of vaccine strain match; and (3) standard-dose vaccines provide demonstrably inferior immunogenicity and clinical protection in adults aged >=65 years due to multifaceted immune decline. Taken together, these findings raise substantive questions about the adequacy and uniformity of the current one-dose-fits-all vaccination framework and suggest that risk-stratified, platform-differentiated, and immunomodulatory approaches deserve rigorous evaluation.
Introduction
Seasonal influenza remains a major cause of morbidity and mortality in the United States, contributing an estimated 9 to 45 million illnesses, up to 810,000 hospitalizations, and 12,000 to 61,000 deaths annually (1). Annual vaccination of all persons aged >=6 months has been recommended by the Advisory Committee on Immunization Practices (ACIP) since 2010, predicated on the assumption that vaccination confers consistent and additive benefit across all age groups and vaccination histories (2).
The scientific foundation for this universal recommendation, however, rests primarily on aggregate VE estimates that obscure considerable heterogeneity by age, subtype, and prior vaccination history. Since 2004, the CDC Influenza Vaccine Effectiveness Networks have estimated adjusted overall VE annually using test-negative case-control designs at sentinel sites across the United States (3). These data reveal that VE has rarely exceeded 60%, has fallen below 20% in at least two seasons, and shows a persistent pattern of lower effectiveness against influenza A(H3N2) compared with A(H1N1) (3,4).
Two host immunological mechanisms have emerged as plausible explanations for this variability: immune imprinting, the phenomenon by which the first influenza exposure in childhood establishes a lifelong immunodominant bias toward that subtype’s antigens (5); and immunosenescence, the progressive, multifactorial deterioration of immune function with aging that reduces both the quantity and quality of adaptive immune responses to vaccination (6). Neither phenomenon is adequately accounted for in current vaccination policy design, recommendations for general populations, or VE communication to clinicians and the public.
This report critically examines both phenomena, evaluates the epidemiological evidence for their influence on measured VE, and considers implications for the premise that universal annual influenza vaccination confers reliably additive public health benefit for all individuals under the current standard-dose inactivated vaccine paradigm.
Methods
We conducted a narrative synthesis of published VE estimates from CDC Influenza Vaccine Effectiveness Networks for the 2004-05 through 2024-25 influenza seasons (3), supplemented by a review of mechanistic and epidemiological literature concerning immune imprinting and immunosenescence published through March 2026. Source databases included PubMed/MEDLINE, CDC MMWR and FluView archives, and the Eurosurveillance sentinel network. Studies were selected for relevance to the specific mechanisms examined; no formal systematic review or meta-analytic protocol was applied. This report reflects the authors’ synthesis and interpretation of publicly available data.
Results
Two Decades of VE Surveillance: Persistent Underperformance
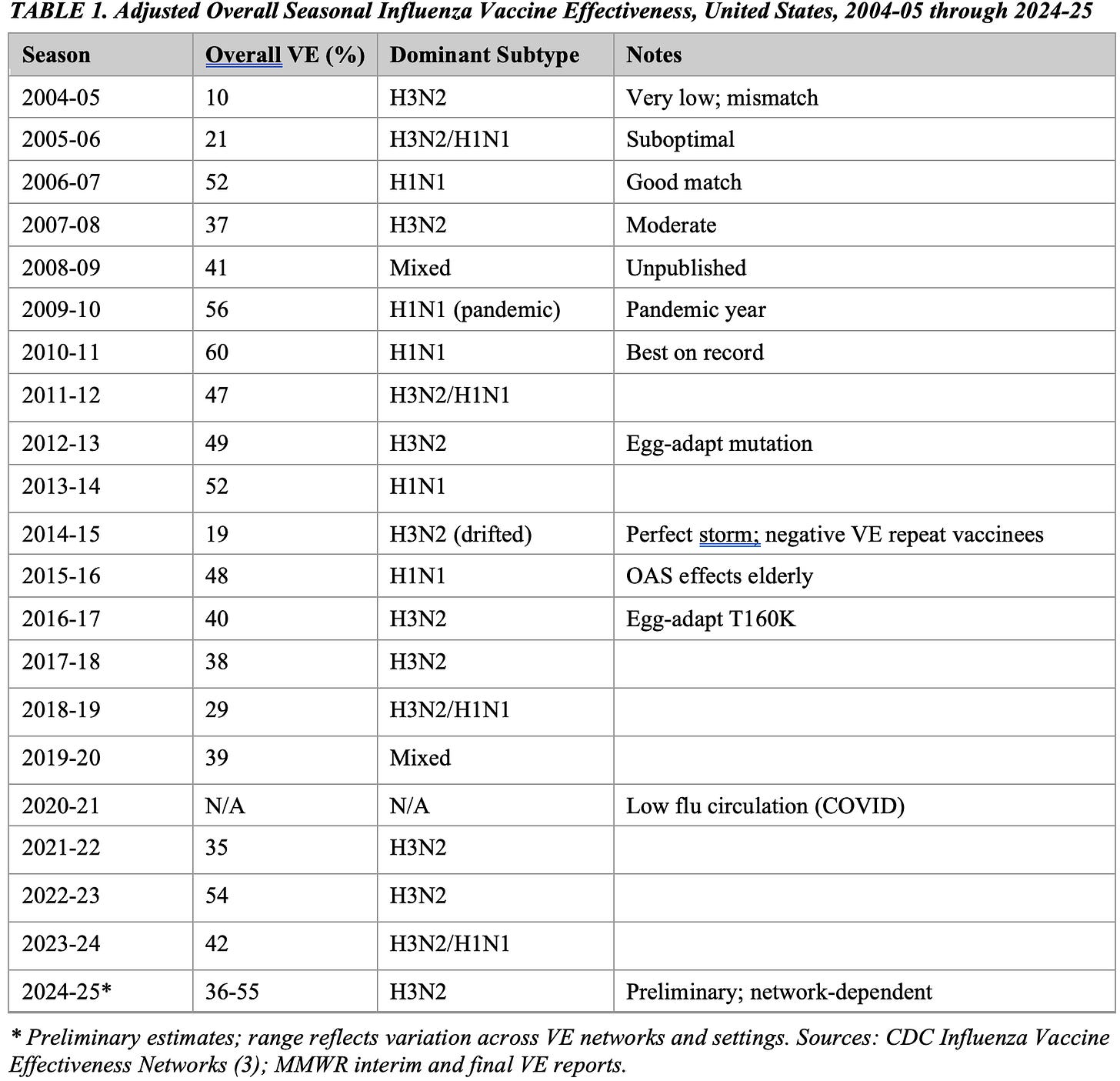
Table 1 summarizes adjusted overall VE estimates from the 2004-05 through 2024-25 influenza seasons. The unweighted mean VE across 20 estimable seasons is approximately 40%, with a range of 10% (2004-05) to 60% (2010-11). No season has achieved VE >=60%, the lower bound of the target range implied by vaccine development standards. The 2020-21 season produced no estimate due to suppressed influenza circulation during the COVID-19 pandemic.
A consistent pattern is evident: seasons dominated by influenza A(H3N2) are associated with lower VE (unweighted mean approximately 35%) compared with seasons dominated by A(H1N1) (unweighted mean approximately 52%). This differential cannot be fully explained by antigenic mismatch alone and is consistent with the hypothesis that immunological factors specific to H3N2 -- including the difficulty of generating broadly neutralizing antibodies against its rapidly evolving hemagglutinin head domain and the influence of birth-cohort imprinting -- systematically depress effectiveness against this subtype (7,8).
Immune Imprinting: Mechanistic Evidence and VE Consequences
The doctrine of original antigenic sin, described by Francis in 1960, proposed that antibody responses to influenza throughout life are dominated by the subtype first encountered in childhood (5). Contemporary immunology has refined this concept into immune imprinting: a lifelong bias in memory B cell and T follicular helper (Tfh) cell repertoires toward the antigens of the first influenza subtype exposure, which preferentially recalls existing memory at the expense of de novo responses to antigenically novel epitopes on drifted strains (9).
The mechanistic basis involves competitive activation dynamics: antigen-experienced memory B cells exhibit higher precursor frequencies, possess enhanced survival programming, and have lower B cell receptor activation thresholds than naive B cells. When a vaccine or infection presents antigens that are structurally similar to the imprinted subtype, memory B cells are preferentially expanded, generating high-titer antibodies against conserved (often suboptimally neutralizing) epitopes and suppressing de novo responses to the strain-specific head epitopes that are the primary neutralization targets (9,10).
Epidemiological evidence for VE-relevant imprinting is substantial. The 2009 A(H1N1) pandemic provided a natural experiment: adults aged >=65 years -- imprinted on H1N1 strains circulating before 1957 -- showed markedly lower infection and mortality rates than younger cohorts imprinted on H3N2 strains, with approximately two thirds demonstrating pre-existing serologic cross-reactivity (11). Conversely, birth cohorts imprinted on H3N2 (born after 1968) experienced disproportionately more severe disease during H1N1-predominant seasons after 2009 (12).
For seasonal VE, the most consistent finding concerns repeat vaccination and H3N2. Skowronski and colleagues, using Canada’s Sentinel Practitioner Surveillance Network with test-negative designs, documented that in the 2014-15 season -- characterized by both an antigenically drifted H3N2 circulating strain and an unchanged vaccine composition from the prior season -- VE in persons with no prior vaccination history was approximately 52%, compared with -32% in those vaccinated in the prior year only and approximately -54% in those vaccinated each year since 2012-13 (13). The investigators described this as a ‘perfect storm’ of antigenic drift, unchanged vaccine composition, and repeat-vaccination immune interference (13).
A subsequent meta-analysis and systematic review by Jones-Gray and colleagues, encompassing 83 studies and 43 meta-analyses, confirmed that consecutive influenza vaccination was associated with attenuated VE for influenza A and B compared with current-season-only vaccination, though the effects were characterized as not severe enough to recommend against annual vaccination (14). A separate Lancet Respiratory Medicine meta-analysis of 41 studies found that vaccination in both the current and prior seasons was associated with lower protection than current-season vaccination alone: delta VE of -9% for A(H1N1), -18% for A(H3N2), and -7% for B (15). The H3N2 attenuation is of particular concern, given that H3N2 already generates the lowest absolute VE values in matched seasons.
The mechanistic framework that unifies these observations -- the antigenic distance hypothesis -- predicts that negative interference from prior vaccination is most likely when the same vaccine strains are used across consecutive seasons but fail to match the current circulating strain. Under these conditions, the prior season’s vaccine imprints memory responses to antigens that are neither identical to the current circulating strain nor to the current season’s vaccine, creating a three-way mismatch that compounds standard antigenic drift effects (16).
A further complication is egg adaptation. Most conventional inactivated influenza vaccines are manufactured in embryonated hens’ eggs, and the hemagglutinin of the H3N2 component in particular undergoes adaptive mutations during this process that alter its antigenic properties relative to the circulating strain (17). If a child’s first influenza exposure is to an egg-adapted vaccine antigen, this imprinting event may adversely shape antibody repertoires for subsequent decades, potentially reducing VE in later life (17). This is particularly relevant now that the WHO recommends vaccination beginning at age 6 months, substantially increasing the proportion of the population whose primary influenza imprint originates from an egg-derived vaccine rather than natural infection.
Immunosenescence: Mechanisms of Vaccine Failure in Older Adults
Adults aged >=65 years account for the overwhelming majority of influenza-related hospitalizations and deaths: during the 2023-24 season, this age group accounted for 51% of hospitalizations and 68% of deaths (18). Yet this is precisely the population in which the standard seasonal influenza vaccine performs most poorly. Immunosenescence -- the progressive, multifactorial deterioration of innate and adaptive immune function with aging -- is the primary biological explanation (6,19).
Immunosenescence is not a single deficit but a convergence of at least five intersecting mechanisms:
Thymic involution and naive T cell depletion. The thymic epithelial space begins declining from the first year of life and contracts at approximately 3% per year until middle age, then at 1% per year thereafter (20). T cell receptor excision circle (TREC) analysis demonstrates a steady three-log decline in circulating naive T cells across an 80-year lifespan (21). By age 65, TCR diversity drops precipitously. Because vaccine responses require de novo priming of naive T cells against new or drifted epitopes, the contracted naive T cell pool limits the breadth and quality of vaccine-induced immunity. Vaccination-induced germinal center Tfh cell clonal lineages have been shown to persist in lymph nodes over multiple years, potentially channeling responses toward historical rather than current antigens (22).
B cell intrinsic defects. Aging reduces both the quantity and functional quality of B cells. Reductions in naive B cell numbers, B cell diversity, germinal center size and frequency in secondary lymphoid organs, and intrinsic B cell effector function all compound to reduce vaccine-induced antibody magnitude and avidity (6,23). High TNF-alpha levels in aged individuals -- a hallmark of inflammaging -- directly downregulate CD28 expression on T and B cells, reducing co-stimulatory signaling required for full B cell activation (24).
Inflammaging. A chronic, sterile, low-grade systemic inflammatory state -- driven by senescent cell accumulation and their senescence-associated secretory phenotype (SASP), defective autophagy, gut dysbiosis, and persistent latent infections -- characterizes aging and is associated with elevated circulating IL-6, TNF-alpha, and CRP (25). Pre-vaccination inflammatory gene signatures are negatively predictive of antibody responses to influenza vaccination in elderly adults (26). While some inflammation is required for vaccine immunogenicity, the chronic basal inflammation of aging appears to occupy signaling pathways in ways that impair rather than augment vaccine-induced adaptive responses.
Innate immune dysfunction and impaired antigen presentation. Age-related declines in toll-like receptor (TLR) engagement -- particularly TLR1/2, TLR3, TLR5, TLR8 in myeloid dendritic cells, and TLR7 and TLR9 in plasmacytoid dendritic cells -- reduce the innate immune sensing that initiates downstream T and B cell activation after vaccination (27). Elevated NK cell populations in older adults have been shown to suppress germinal center responses, providing an additional brake on vaccine-induced humoral immunity (28).
CD8 cytotoxic T cell failure. Standard inactivated influenza vaccines generate a weak stimulus to the cytotoxic T lymphocyte (CTL) arm of adaptive immunity, relying primarily on re-stimulation of pre-existing CD8 memory rather than de novo priming (27). Age-related decline in CTL generation and function -- driven by impaired CD4 T helper cell co-stimulation, reduced IL-2 signaling, and exhaustion phenotypes in persisting memory cells -- directly reduces the capacity to clear influenza virus from the lower respiratory tract, which is the critical determinant of severe disease prevention (27).
The aggregate consequence is that standard-dose vaccines produce demonstrably inferior immunogenicity in older adults by all conventional measures -- hemagglutinin inhibition (HAI) titers, seroconversion rates, and antibody avidity -- and that epidemiological VE estimates confirm lower protection against both outpatient illness and hospitalization (6,19). Critically, the mechanisms driving immunosenescence overlap with those through which immune imprinting operates: both involve the dominance of antigen-experienced memory cells over naive B cell responses, and both are exacerbated by repeat vaccination that back-boosts established memory while suppressing de novo antibody generation (9,22).
Enhanced Vaccine Formulations: Partial Mitigation and Unresolved Questions
Three vaccine strategies have been developed specifically to address immunosenescence in adults aged >=65 years: high-dose inactivated vaccine (HD-IIV, containing four times the standard antigen dose), MF59-adjuvanted inactivated vaccine (aIIV), and recombinant hemagglutinin vaccine (RIV). ACIP issued a preferential recommendation for these enhanced formulations over standard-dose vaccine in this age group beginning with the 2022-23 season (2).
A meta-analysis of 12 influenza seasons and over 45 million individuals aged >=65 years demonstrated that HD-IIV provided significantly better protection than standard-dose vaccine against influenza-like illness, influenza-related hospitalizations, and cardiovascular outcomes (29). Real-world Kaiser Permanente data from the 2022-23 season found that, compared with standard-dose egg-based vaccine, the relative effectiveness against PCR-confirmed hospitalization was 25% for HD-IIV and 62% for aIIV (30). These represent clinically meaningful incremental gains above a low baseline.
However, recent randomized controlled trial evidence complicates this picture. The DANFLU-2 trial, the largest individually randomized trial of HD versus standard-dose vaccine conducted to date (n = 332,438 Danish adults aged >=65 years across three influenza seasons, 2022-23 through 2024-25), found that HD-IIV did not result in a significantly lower incidence of hospitalization for influenza or pneumonia compared with standard-dose vaccine (relative VE 5.9%; 95% CI -2.1 to 13.4; p = 0.14) (31). The authors noted this finding was inconsistent with prior observational and randomized data, potentially reflecting contextual factors including circulating strain composition and background immunity.
Pharmacological strategies targeting the hallmarks of aging prior to vaccination are an emerging frontier. Administration of the mTOR inhibitor RAD001 (everolimus) before influenza vaccination in adults aged >=65 years increased antibody titers against all three vaccine strains by more than 20% and reduced proportions of T cells expressing the exhaustion marker PD-1 (33). Metformin, which reduces circulating CRP, IL-6, and TNF-alpha, has been associated with higher post-vaccination antibody titers in diabetic patients compared with other hypoglycemics (33). These approaches remain investigational.
Discussion
The evidence reviewed in this report converges on a conclusion that is uncomfortable in its public health implications: the current universal seasonal influenza vaccination policy, premised on a standard-dose inactivated vaccine given annually to virtually the entire population, does not perform as a uniformly effective intervention. For a substantial proportion of vaccinated individuals -- particularly older adults receiving their fifth, tenth, or fifteenth consecutive annual vaccination, individuals born in birth cohorts imprinted on influenza A subtypes that diverge from the dominant circulating strain, and individuals receiving standard-dose vaccines during H3N2-predominant seasons -- the measured protection is modest at best and, in certain epidemiological configurations, may be functionally negligible.
This assessment does not imply that influenza vaccination is without benefit. Even in the worst-performing seasons, vaccination is associated with reductions in ICU admission and influenza-attributable mortality compared with no vaccination (4). Enhanced formulations provide meaningful additional protection in older adults over standard-dose vaccines, though the magnitude of this benefit may be smaller and more variable than the observational literature suggested (29-31). The argument here is not against vaccination per se but against the intellectual and policy complacency embedded in a universal recommendation that has not been substantially revised to account for the immune biology of those it aims to protect.
Three specific policy gaps follow from the evidence reviewed:
The repeat vaccination paradox. Current policy encourages and in some contexts requires annual vaccination without regard to prior vaccination history. The evidence from multiple independent VE studies and meta-analyses indicates that prior-year vaccination attenuates VE, particularly against H3N2 in mismatch seasons, by an average of approximately 18-20% in the most rigorous estimates (14,15). This is not a marginal finding. In a season such as 2014-15, the interaction between an unchanged vaccine composition, antigenic drift in H3N2, and repeat vaccination produced what investigators described as near-zero or negative VE in multiply vaccinated individuals (13). The mechanism -- back-boosting of outdated memory B cell responses at the expense of de novo neutralizing antibody generation -- is biologically coherent and supported by both animal model and human immunogenicity data (9,10). Public health guidance that does not acknowledge this attenuation effect misleads clinicians and patients about the expected benefit of vaccination.
The immunosenescence gap. Adults aged >=65 years bear the largest absolute burden of influenza mortality yet derive the least reliable protection from standard-dose vaccines, due to thymic involution, B cell dysfunction, inflammaging, and innate immune impairment (6,19). The ACIP preferential recommendation for enhanced formulations in this age group is a recognition of the problem; however, the evidence base for the effectiveness of these formulations against severe outcomes such as hospitalization and death -- the outcomes that matter most -- remains uncertain, as the DANFLU-2 trial results demonstrate (31). Furthermore, even if enhanced vaccines modestly improve immunogenicity, the fundamental biology of immunosenescence means that seroconversion rates and HAI titers generated by senescent immune systems may not faithfully predict clinical protection in the way they do in younger adults (32). The correlates of protection may differ.
The imprinting-policy disconnect. Current vaccination policy is strain-agnostic with respect to the recipient’s birth cohort and prior immune history. Yet the evidence -- from pandemic epidemiology, from birth-cohort-specific VE analyses, and from mechanistic immunology -- indicates that an individual’s first influenza exposure in childhood is a major determinant of their subsequent vaccine response for decades (5,9,11). In particular, the large proportion of the current elderly population in the United States that was imprinted on H1N1 strains circulating before 1968 faces both immunosenescence-related immune decline and imprinting-related suboptimal responses to H3N2 vaccine antigens -- a compounding of disadvantages that the current policy framework does not address.
Taken together, these considerations suggest that the scientific justification for strict universal annual vaccination -- as opposed to risk-stratified, platform-differentiated, or historically informed approaches -- is weaker than is commonly represented. This does not diminish the importance of protecting high-risk populations with the best available tools. It does argue for investment in more targeted strategies: universal use of enhanced formulations in adults aged >=65 years, accelerated evaluation of mRNA vaccines and their effectiveness against severe outcomes, clinical trial evaluation of immunomodulatory pre-treatment to address inflammaging, and policy-level acknowledgment that the benefit of annual vaccination in previously multiply-vaccinated individuals in matched versus mismatched H3N2 seasons may differ substantially from the headline VE figures communicated to the public.
This report has limitations. VE estimates from test-negative designs are observational and subject to confounding by indication and frailty bias, although standard adjustment procedures mitigate but do not eliminate these effects. The mechanistic evidence for immune imprinting’s effects on VE derives partly from observational birth-cohort analyses and animal models; the degree to which childhood imprinting modifies individual-level VE is not yet quantifiable with precision. The evidence base for immunosenescence mechanisms is robust in terms of cellular biology but translating cellular observations to population-level VE is imprecise. Nevertheless, the convergence of mechanistic plausibility with consistent epidemiological patterns across multiple independent surveillance systems in multiple countries provides a foundation for policy reconsideration that has been insufficiently engaged.
Conclusions
Two decades of influenza VE surveillance reveal a consistently underperforming vaccine whose limitations are substantially attributable to host immunological factors -- immune imprinting and immunosenescence -- rather than antigenic mismatch alone. Repeat annual vaccination attenuates VE against influenza A(H3N2) through immunological interference with de novo antibody responses; older adults, the primary target of influenza mortality reduction efforts, receive the least reliable protection from the standard platform recommended for the general population; and the combination of childhood subtype imprinting and age-related immune decline in the current elderly cohort creates compounding vulnerabilities that existing policy does not address. Enhanced vaccine formulations provide partial mitigation, but their superiority over standard-dose vaccines against severe outcomes is not firmly established by randomized trial evidence as of 2025. Universal seasonal influenza vaccination remains a valuable public health intervention, but the homogeneity of current policy design is misaligned with the heterogeneity of host immune biology. Risk-stratified recommendations, accelerated development and evaluation of next-generation vaccine platforms, and transparent public communication about the conditional nature of annual vaccination benefit are warranted.
Acknowledgments
The author acknowledges the ongoing contributions of the CDC Influenza Vaccine Effectiveness Networks and their academic collaborators, who have generated the surveillance data underlying this analysis. No funding specific to this review was received. The author formerly served as the vice chairperson of the CDC ACIP committee and chairperson of the influenza vaccine work group, but consequent to a recent court decision in the case of AAP vs HHS, no longer has any affiliation with the CDC or the ACIP. The views expressed are those of the author alone, and do not reflect the opinions of the USG, HHS, CDC, or ACIP.
Conflict of Interest Statement
No conflicts of interest are reported.
References
1. Rolfes MA, Foppa IM, Garg S, et al. Annual estimates of the burden of seasonal influenza in the United States: a tool for strengthening influenza surveillance and preparedness. Influenza Other Respir Viruses. 2018;12(1):132-137. https://doi.org/10.1111/irv.12486
2. Grohskopf LA, Blanton LH, Ferdinands JM, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices -- United States, 2022-23 influenza season. MMWR Recomm Rep. 2022;71(1):1-28. https://doi.org/10.15585/mmwr.rr7101a1
3. Centers for Disease Control and Prevention. CDC seasonal flu vaccine effectiveness studies. Accessed March 2026. https://www.cdc.gov/flu-vaccines-work/php/effectiveness-studies/index.html
4. Rolfes MA, Flannery B, Chung JR, et al. Effects of influenza vaccination in the United States during the 2017-2018 influenza season. Clin Infect Dis. 2019;69(11):1845-1853. https://doi.org/10.1093/cid/ciz075
5. Francis T Jr. On the doctrine of original antigenic sin. Proc Am Philos Soc. 1960;104(6):572-578.
6. Dugan HL, Henry C, Wilson PC. Aging and influenza vaccine-induced immunity. Cell Immunol. 2020;348:103998. https://doi.org/10.1016/j.cellimm.2019.103998
7. Belongia EA, Simpson MD, King JP, et al. Variable influenza vaccine effectiveness by subtype: a systematic review and meta-analysis of test-negative design studies. Lancet Infect Dis. 2016;16(8):942-951. https://doi.org/10.1016/S1473-3099(16)00129-6
8. Cobey S, Hensley SE. Immune history and influenza virus susceptibility. Curr Opin Virol. 2017;22:105-111. https://doi.org/10.1016/j.coviro.2016.12.004
9. King SM, Bryan SP, Hilchey SP, Wang J, Zand MS. First impressions matter: immune imprinting and antibody cross-reactivity in influenza and SARS-CoV-2. Pathogens. 2023;12(2):169. https://doi.org/10.3390/pathogens12020169
10. Henry C, Palm AKE, Krammer F, Wilson PC. From original antigenic sin to the universal influenza virus vaccine. Trends Immunol. 2018;39(1):70-79. https://doi.org/10.1016/j.it.2017.08.003
11. Adalja AA, Toner E, Inglesby TV. Original antigenic sin and pandemic (H1N1) 2009. Emerg Infect Dis. 2010;16(6):1028-1029. https://doi.org/10.3201/eid1606.091653
12. Gostic KM, Ambrose M, Worobey M, Lloyd-Smith JO. Potent protection against H5N1 and H7N9 influenza via childhood hemagglutinin imprinting. Science. 2016;354(6313):722-726. https://doi.org/10.1126/science.aag1322
13. Skowronski DM, Chambers C, Sabaiduc S, et al. A perfect storm: impact of genomic variation and serial vaccination on low influenza vaccine effectiveness during the 2014-2015 season. Clin Infect Dis. 2016;63(1):21-32. https://doi.org/10.1093/cid/ciw176
14. Jones-Gray E, Robinson EJ, Kucharski AJ, Fox A, Sullivan SG. Does repeated influenza vaccination attenuate effectiveness? A systematic review and meta-analysis. Lancet Respir Med. 2023;11(1):27-44. https://doi.org/10.1016/S2213-2600(22)00266-1
15. McLean HQ, Thompson MG, Sundaram ME, et al. Impact of repeated vaccination on vaccine effectiveness against influenza A(H3N2) and B during 8 seasons. Clin Infect Dis. 2014;59(10):1375-1385. https://doi.org/10.1093/cid/ciu680
16. Skowronski DM, Chambers C, De Serres G, et al. Serial vaccination and the antigenic distance hypothesis: effects on influenza vaccine effectiveness during A(H3N2) epidemics in Canada, 2010-2011 to 2014-2015. J Infect Dis. 2017;215(7):1059-1099. https://doi.org/10.1093/infdis/jiw605
17. Gouma S, Anderson EM, Hensley SE. The impact of egg adaptation and immune imprinting on influenza vaccine effectiveness. Vaccine. 2025. https://doi.org/10.1016/j.vaccine.2025.126905
18. Centers for Disease Control and Prevention. FluView: influenza-associated hospitalizations. Accessed March 2026. https://www.cdc.gov/flu-burden/
19. Poland GA, Ovsyannikova IG, Kennedy RB. Personalized vaccinology: a review. Vaccine. 2018;36(36):5350-5357. https://doi.org/10.1016/j.vaccine.2017.07.062
20. Palmer DB. The effect of age on thymic function. Front Immunol. 2013;4:316. https://doi.org/10.3389/fimmu.2013.00316
21. Hakim FT, Gress RE. Thymic involution and immune reconstitution. Trends Immunol. 2009;30(7):300-306. https://doi.org/10.1016/j.it.2009.03.008
22. Schattgen SA, Guion K, Crawford JC, et al. Linking T cell receptor sequence to functional phenotype at the single-cell level. Nat Immunol. 2024;25:131-144. Cited in: Herath TK, et al. Vaccination against influenza viruses annually: renewing or narrowing the protective shield? J Exp Med. 2025;222(7):e20241283.
23. Frasca D, Blomberg BB. Aging and vaccines: the use of system biology approaches for developing vaccines for the aging population. Exp Gerontol. 2014;53:62-67. https://doi.org/10.1016/j.exger.2013.12.010
24. Frasca D, Diaz A, Romero M, Landin AM, Blomberg BB. High TNF-alpha levels in resting B cells negatively correlate with their response. Exp Gerontol. 2014;54:116-122. https://doi.org/10.1016/j.exger.2014.01.004
25. Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15(9):505-522. https://doi.org/10.1038/s41569-018-0064-2
26. Nakaya HI, Wrammert J, Lee EK, et al. Systems biology of vaccination for seasonal influenza in humans. Nat Immunol. 2011;12(8):786-795. https://doi.org/10.1038/ni.2067
27. McElhaney JE, Andrew MK, McNeil SA. Estimating influenza vaccine effectiveness: evolution of methods to better understand the confounding effects of frailty. Vaccine. 2017;35(49 Pt B):6989-6996. https://doi.org/10.1016/j.vaccine.2017.10.018
28. Cossarizza A, Ortolani C, Forti E, et al. NK cells and influenza: non-specific effectors or immunoregulators? J Leukoc Biol. 2004;76(4):791-798.
29. Lee JKH, Lam GKL, Shin T, et al. High-dose influenza vaccine in older adults by age and seasonal characteristics: systematic review and meta-analysis update. Vaccine X. 2023;14:100327. https://doi.org/10.1016/j.jvacx.2023.100327
30. Ku JH, Rayens E, Sy LS, et al. Comparative effectiveness of adjuvanted and high-dose versus standard-dose influenza vaccines against influenza-related medical encounters and hospitalizations in older adults, 2022-2023. Clin Infect Dis. 2024. https://doi.org/10.1093/cid/ciae407
31. Johansen ND, Skaarup KG, Biering-Sorensen T, et al. High-dose influenza vaccine effectiveness against hospitalization in older adults: the DANFLU-2 randomized trial. N Engl J Med. 2025. https://doi.org/10.1056/NEJMoa2509907
32. Haddad F, Dokmak G, Karaman R. Humoral immune response to mRNA-based influenza vaccines in older adults: a systematic review and meta-analysis of randomized controlled trials. PMC. 2025.
33. Camacho-Muñoz D, et al. Targeting the hallmarks of aging to improve influenza vaccine responses in older adults. Immun Ageing. 2023;20:22. https://doi.org/10.1186/s12979-023-00348-6


Interestingly, what I find most disturbing is reading about your former ACIP involvement. I know something will change in regard to this current state, but the unknown outcome is devastating. And this research is invaluable information and recommendation of change of the vaccine for the elderly.
Dr. Malone, thanks very much for putting all this information together in a clear concise manner. I try to stay informed about issues that could affect my health, mid 70s male with respiratory problems, and articles like this are very helpful. Your work is very much appreciated.