Why your flu shot may work differently than you think
Two decades of federal surveillance data reveal how immune imprinting and an aging immune system undermine flu vaccine effectiveness.
Why your flu shot may work differently than you think
Two decades of federal surveillance data reveal how immune imprinting and an aging immune system undermine flu vaccine effectiveness. Current universal vaccination policy has not caught up to the biology.
Every autumn, public health authorities deliver a message that is simultaneously accurate and misleading: get your flu shot. All Americans aged six months and older are recommended to receive one. The guidance is consistent, reassuring, and considerably more complicated than it sounds.
A synthesis of two decades of CDC surveillance data, which will be published separately in Malone.News makes an argument that has been quietly building in the immunology literature for years: the current one-size-fits-all approach to seasonal influenza vaccination fails to account for two fundamental features of how the immune system actually works. The first is that your first flu exposure as a child permanently shapes how you respond to every flu vaccine you will ever receive. The second is that the immune system ages in ways that make older adults both the most important target for vaccination and the least reliable responders to it.
This is not a case against flu vaccination. It is a case for being honest about what the vaccine does and does not do, and for whom. In other words, it is a case for open and transparent informed consent.
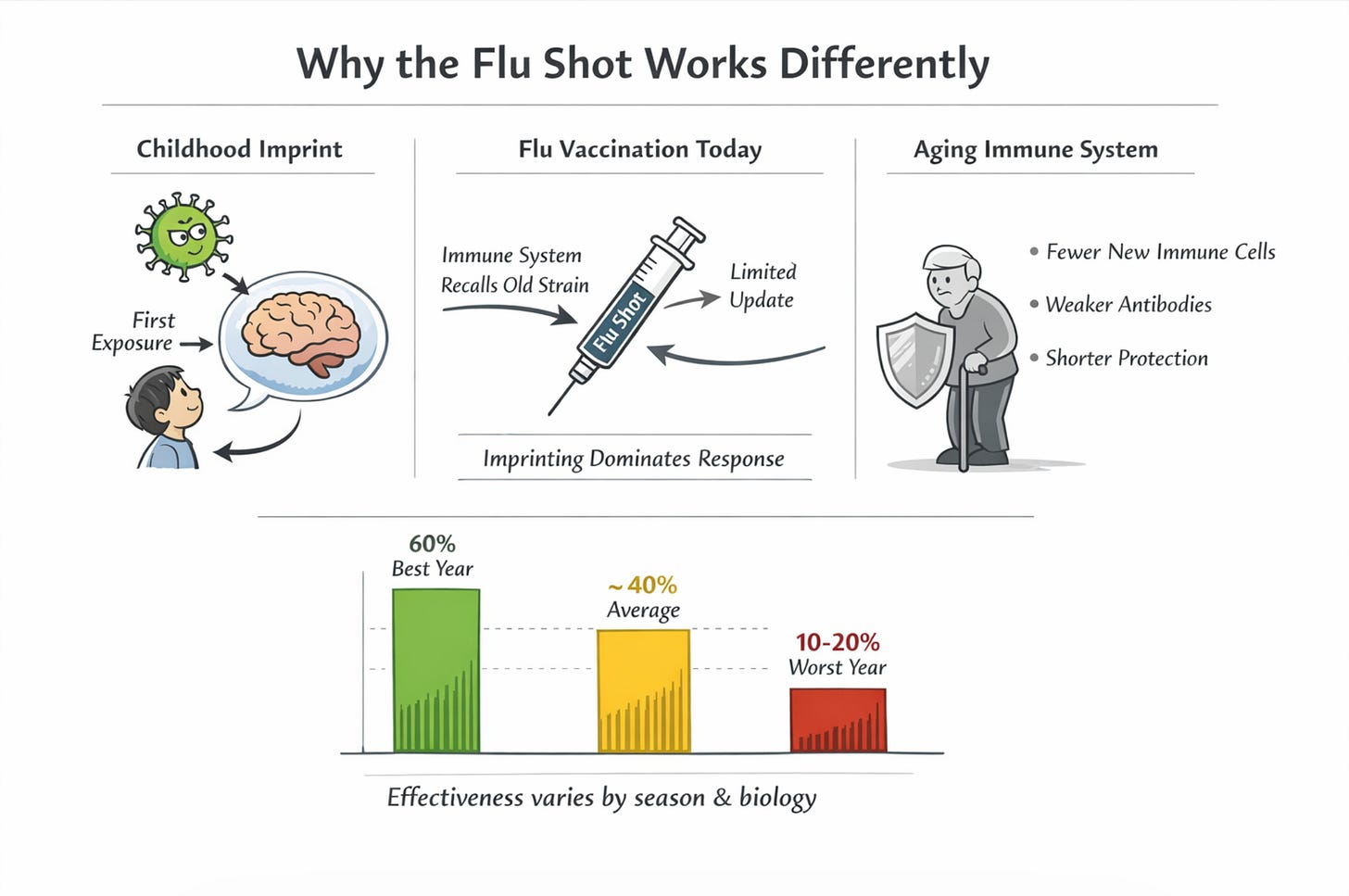
The track record: never reliably above 60%
Before getting to the biology, it is worth establishing how variable the vaccine’s performance actually is. Since 2004, the CDC has estimated seasonal flu vaccine effectiveness every year through a network of sentinel clinics and hospitals. The results tell a different story than the usual reassuring headlines and approved narratives.
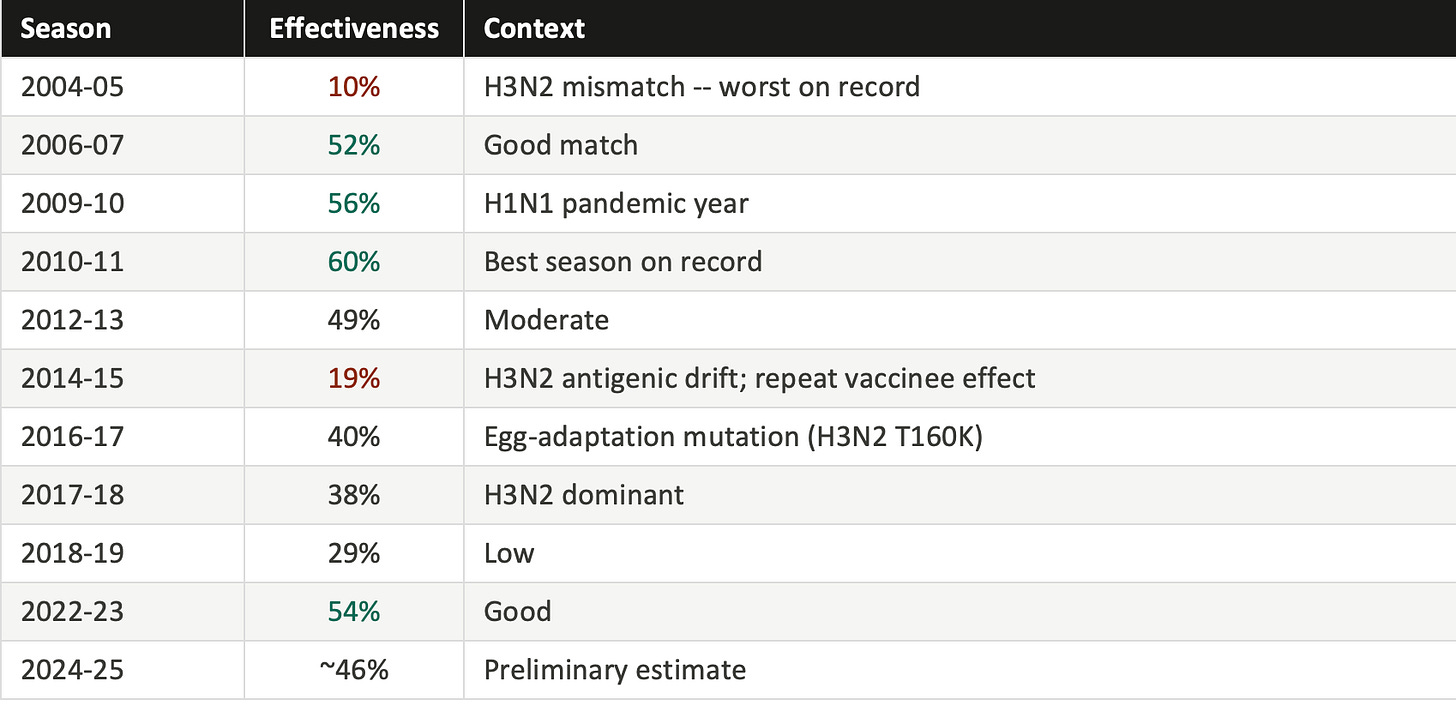
Vaccine effectiveness by season (selected years)
Source: CDC Influenza VE Networks, 2004-2026.
A 40% average effectiveness figure means that roughly four in ten flu cases that would have occurred among unvaccinated people were prevented among vaccinated people. That is genuinely useful. But it is far below vaccines like measles (97% effective) or childhood polio (99% effective). And in bad years, it falls into territory where the protection is marginal.
The culprit in most low-effectiveness years is the H3N2 subtype of influenza A. It evolves faster than the other major circulating strains, it is harder to match, and it tends to mutate in ways that are specifically worsened by the egg-based manufacturing process most flu vaccines still rely on. In H3N2-dominant years, which occur in roughly a third to half of recent seasons, effectiveness against that specific strain has sometimes fallen below 20%.
These fluctuations are not random noise. They have patterns, and those patterns trace back to biology that current policy largely ignores.
The ghost of flu seasons past: immune imprinting
In 1960, a virologist named Thomas Francis Jr. described something he called “original antigenic sin.” When people were vaccinated against influenza strains they had never seen before, their immune response was still dominated by the flu virus they had first encountered as children, sometimes decades earlier. Like a palimpsest (an overwritten manuscript), the old exposure kept showing through.
Modern immunology has renamed this immune imprinting and clarified the mechanism in considerable detail. The immune system maintains a permanent library of memory cells -- B cells and T cells -- tuned to pathogens it has encountered before. These memory cells vastly outnumber naive cells; they activate faster and respond more vigorously. When a new vaccine arrives with antigens that are similar but not identical to a past exposure, the memory cells are preferentially activated. The immune system, essentially, recalls the old answer rather than generating a new one.
THE 2009 PANDEMIC MADE THIS VISIBLE IN REAL TIME
When the H1N1 pandemic emerged in 2009, illness patterns did not follow age lines as expected. They followed birth-year lines. Adults over 65, whose childhoods occurred during a period when H1N1 strains dominated, were largely protected. About two thirds had pre-existing immunity from exposures that had occurred decades earlier. Middle-aged adults, whose early flu experiences were dominated by H3N2 strains circulating since 1968, were the most vulnerable. Their immune memory was tuned to the wrong flu family. The same vaccine given to both groups produced different levels of protection depending on what each person’s immune system had memorized as a child.
The immune system tends to recall the old answer rather than generating a new one -- even when the old answer is subtly wrong.
A series of Canadian surveillance studies conducted between 2012 and 2019 documented this phenomenon with striking clarity. In the 2014-15 season, a year when the dominant circulating H3N2 virus had drifted significantly from the vaccine strain while the vaccine composition remained unchanged from the prior year, people who had been vaccinated in the previous one to two years showed near-zero or even negative effectiveness against the dominant strain. A systematic review published in the Lancet Respiratory Medicine in 2023, analyzing 83 studies, confirmed that consecutive annual vaccination is associated with approximately 18 to 20% lower effectiveness against H3N2 compared to vaccinating in the current season alone.
The data indicate that vaccination still protected better than no vaccination. But the accumulating immune imprint from prior exposures measurably reduced the precision of the response. The immune system, having been repeatedly told to remember one answer, struggled to learn a new one.
THE MECHANISM
Memory cells win the competition
When vaccine antigens arrive, memory B cells trained on past exposures activate within hours. Naive B cells, which would generate genuinely updated antibodies, need days to weeks to respond. In an established immune system, they are effectively outcompeted. The result is a response tuned to the past rather than the present flu season.
THE EGG PROBLEM
Manufacturing introduces another mismatch
Most flu vaccines are still grown in chicken eggs. The H3N2 virus adapts to avian cells during this process, subtly shifting its surface proteins away from those that circulate in humans. A child vaccinated with an egg-adapted antigen may carry a slightly misaligned immune imprint for decades, a consequence of the production method encoded in immune memory from the very first vaccination.
When the immune system ages out: immunosenescence
The people who most need the flu vaccine -- adults over 65, who account for more than two thirds of flu deaths every year -- are also the people whose immune systems respond to it least reliably. Understanding why requires a brief look at how the immune system changes with age.
Four converging changes
FROM BIRTH Thymic involution: the training facility closes
The thymus, the organ where new T cells are trained and released into circulation, begins shrinking from the first year of life. By the mid-60s, T cell output has fallen by roughly 95% from its peak. The pool of naive T cells available to mount a fresh response to a new vaccine is drastically reduced. The immune system becomes increasingly dependent on its existing memory library, which, as described above, can work against precision.
BY AGE 50 Inflammaging: a persistent low-grade fire
As we age, a chronic, sterile, low-grade inflammatory state develops, driven by senescent cells accumulating in tissue, gut microbiome changes, and persistent low-level infections. Blood levels of inflammatory markers like IL-6, TNF-alpha, and CRP rise. This state, called inflammaging, appears to occupy immune signaling pathways in ways that blunt vaccine responses, even as it reflects immune system activity. Some inflammation is necessary for vaccines to work, but the chronic background inflammation of aging occupies channels without providing the coordinated response that generates protection.
BY AGE 65 Germinal center decline: the antibody factory closes
Lymph nodes contain structures called germinal centers, which are microscopic factories where B cells compete to produce better and better antibodies through a process of mutation and selection. With age, lymph nodes shrink, germinal centers become fewer and less active, and the quality of the antibodies produced decreases. Antibodies generated in older immune systems are fewer in number, bind their targets less tightly, and wane faster. This is why the same vaccine that generates robust, lasting immunity in a 30-year-old may generate a weaker, shorter-lived response in an 80-year-old.
ONGOING The compounding interaction
Immunosenescence and immune imprinting reinforce each other. As the naive T and B cell pool shrinks, the immune system becomes even more reliant on existing memory, which intensifies the imprinting effect. An 80-year-old’s immune response to this year’s flu vaccine is more dominated by childhood exposures than a 40-year-old’s. The two phenomena together create a compounding disadvantage that is most acute in the people who need protection most.
A COUNTERINTUITIVE POINT
Lower side effects in older adults after a flu shot are not a sign that the vaccine is working well; often, it is the opposite. The same immune dysfunction that reduces the protective response also reduces the inflammatory response that produces arm soreness, fatigue, and low-grade fever. An 80-year-old who feels nothing after a flu shot is not necessarily better protected than a 35-year-old who feels lousy for a day. Reactogenicity is a rough proxy for immune activation, and both go down together with immunosenescence.
Three gaps in current policy
The scientific review examined here identifies three specific ways that current universal vaccination policy fails to account for what the biology shows.
1. The repeat vaccination paradox
Current policy encourages and, in some settings, effectively requires annual vaccination without any consideration of prior vaccination history. But the evidence shows that in H3N2-mismatched seasons, which occur frequently, prior-year vaccination can reduce effectiveness by an average of 18 to 20%. The mechanism involves back-boosting of outdated immune memory that crowds out a fresh response. This effect is biologically coherent and has been replicated across multiple independent surveillance systems in multiple countries. Yet patients are never informed that their vaccination history may influence how well this year’s shot works. The scientific literature acknowledges this effect; public health guidance does not.
2. The immunosenescence gap in older adults
Adults over 65 bear 68% of flu deaths yet receive the least reliable protection from standard-dose vaccines. The U.S. Advisory Committee on Immunization Practices made an important step forward in 2022 by preferentially recommending enhanced formulations, specifically high-dose, MF59-adjuvanted, or recombinant vaccines, for this group. These do meaningfully produce better immune responses. But even the most rigorously conducted clinical trial testing a high-dose vaccine against a standard-dose vaccine, enrolling 332,000 older Danish adults over three seasons (DANFLU-2, published 2025), found no statistically significant difference in hospitalization rates. The fundamental biology of immunosenescence means that improving immunogenicity (the number of antibodies generated) does not automatically translate into improved clinical protection. The correlates of immunity may operate differently in aging immune systems than in younger ones.
3. The imprinting-policy disconnect
Every vaccination decision is made without reference to the recipient’s birth cohort or immune history, specifically what flu strain they first encountered as a child and which family of viruses their immune system is fundamentally tuned to respond to. Yet the evidence from pandemic epidemiology, from birth-cohort-specific effectiveness analyses, and from mechanistic immunology all converge on the same conclusion: that first childhood flu exposure is a major lifelong determinant of vaccine response. The current elderly cohort in the United States was largely imprinted on H1N1 strains circulating before 1957. These individuals face not only immunosenescence but also suboptimal responses to H3N2 vaccine components, a compounding of disadvantages that the current policy framework neither acknowledges nor addresses.
What this does and does not mean
It is worth being precise about the argument here. Even in years with poor effectiveness, current data indicate that vaccination reduces ICU admission and flu-attributable mortality compared to no vaccination. The benefit for adults over 75, where the number needed to vaccinate to prevent one hospitalization is approximately 390 to 540 in a well-matched season, is genuine and clinically meaningful. Enhanced formulations for older adults provide real additional benefit over standard-dose vaccines. None of this is contested.
What is contested is the framing. The aggregate “40 to 60% effective” figure communicated to the public obscures genuine variation by age, birth cohort, prior vaccination history, and season type. Someone who has been vaccinated for 15 consecutive years is in a measurably different immunological position than someone being vaccinated for the first time. An 80-year-old is not in the same position as a 40-year-old, even if they receive the same product on the same day at the same pharmacy.
The path forward requires two things that tend to advance together: more honest communication about what the current vaccines do and for whom, and investment in platforms capable of overcoming the biological obstacles that current vaccines cannot overcome.
Cell-based and recombinant platforms already avoid the egg-adaptation problem that has contributed to H3N2 underperformance. Adjuvanted vaccines and pre-vaccination immunomodulation, including mTOR inhibitors that have shown a 20% improvement in vaccine response in older adults in clinical trials, represent a serious near-term research frontier.
None of these solutions are ready to replace the existing approach tomorrow. But the existing approach has not exceeded 60% effectiveness in 20 years of tracking. At some point, describing a structural limitation as a weather problem -- an unfortunate mismatch this particular season -- becomes a way of avoiding a harder conversation about the platform itself.
Sources and further reading
CDC Influenza Vaccine Effectiveness Networks, season-by-season effectiveness estimates 2004-2026. cdc.gov/flu-vaccines-work/php/effectiveness-studies/
Skowronski DM, Chambers C, Sabaiduc S, et al. A perfect storm: impact of genomic variation and serial vaccination on low influenza vaccine effectiveness during the 2014-2015 season. Clinical Infectious Diseases. 2016;63(1):21-32.
Jones-Gray E, Robinson EJ, Kucharski AJ, Fox A, Sullivan SG. Does repeated influenza vaccination attenuate effectiveness? A systematic review and meta-analysis. Lancet Respiratory Medicine. 2023;11(1):27-44.
Henry C, Palm AKE, Krammer F, Wilson PC. From original antigenic sin to the universal influenza virus vaccine. Trends in Immunology. 2018;39(1):70-79.
Dugan HL, Henry C, Wilson PC. Aging and influenza vaccine-induced immunity. Cellular Immunology. 2020;348:103998.
Herath TK, et al. Vaccination against influenza viruses annually: renewing or narrowing the protective shield? Journal of Experimental Medicine. 2025;222(7):e20241283.
Gostic KM, Ambrose M, Worobey M, Lloyd-Smith JO. Potent protection against H5N1 and H7N9 influenza via childhood hemagglutinin imprinting. Science. 2016;354(6313):722-726.
Johansen ND, et al. High-dose influenza vaccine effectiveness against hospitalization in older adults: the DANFLU-2 randomized trial. New England Journal of Medicine. 2025. doi:10.1056/NEJMoa2509907
This article summarizes findings from a peer-reviewed-style scientific synthesis. It does not constitute medical advice. Speak with a qualified healthcare provider about your individual vaccination decisions.



I’ve never had a flu shot. Don’t believe in them. I rarely get sick. I have a regiment of vitamins and supplements I take daily and eat whole organic foods. I don’t exercise to the extreme but will walk with my wife and the mutts. I tend to my small garden in the summers and do projects around the house. I get a good sweat on at least once a week if not more and I’m not changing what ain’t broke.
Is there a chart of birth years and flu type exposure? Refusing all shots based on covid doesn't appear smart in the older population, and it seems you're suggesting 65+ may benefit from it. As someone who has received a single flu shot in my life, using instead a single high dose vitamin A oral dose has kept me flu free. Enormous vaccine hesitancy here!